Big step in rapid diagnostics for infectious diseases
The speedy, high-tech method of inexpensive, accurate and high-throughput protein biomarker assay testing is being touted as a much-needed development in point-of-care (PoC) testing, say U.S. and Flinders University researchers who specialize in "green" vortex fluidic device (VFD) medical applications.
The new method—using the revolutionary VFD developed by Flinders University—can be readily scaled up to test hundreds and "potentially thousands" of proteins in one assay in less than five minutes, says senior researcher University of California, Irvine Professor Gregory Weiss, who is a U.S. leader in VFD experiments and applications.
"Most importantly, the data produced can be accurately read with a cell phone camera—immediately addressing the gap between development and implementation of biomarker-based precision medicine," he says.
While many costly tests can take upwards of 48–72 hours, on-the-spot tests such as this can lead to accurate diagnosis and early commencement of important drug treatments and reduction of wrong medications.
An accessible universal solution to offer rapid personalized mobile testing for infectious diseases—or diagnosis and delivery of targeted treatments for other diseases even in remote areas—has so far been out of reach.
"The extreme disparity between technologically lagging and advanced settings directly impacts disease mortality and morbidity, particularly for infectious diseases," says Flinders University researcher Dr. Xuan Luo, a co-author in a new article on the novel test.
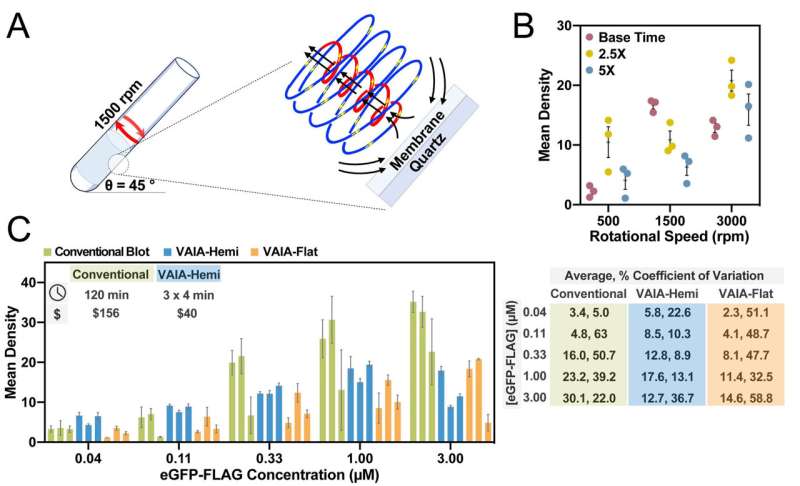
In the assessment published in international chemistry journal Angewandte Chemie, the new method, labeled VFD-accelerated immunoblot assay (VAIA), was compared and found superior to other PoC biomarker-based tests currently in use.
Not only was the processing faster and accurate, it sliced up to 70% off the cost of sometimes toxic reagents. While many immunoblot assays are used to detect certain diseases, many are not highly sensitive and complex to use—even for technicians in a laboratory or clinical setting.
In contrast the VAIA was found to improve conventional processing time from hours to less than five minutes, using three major immunoassay formats with purified proteins and biofluids.
More information: Emily C. Sanders et al, Under‐5‐Minute Immunoblot Assays by Vortex Fluidic Device Acceleration, Angewandte Chemie International Edition (2022). DOI: 10.1002/anie.202202021




















