COVID-19 vaccine developed by Brazilian scientists is ready for clinical trials
Testing on humans of a novel COVID-19 vaccine developed in Brazil will begin this year. It performed well in animal trials, as reported in an article published in August in Nature Communications. The scientists responsible for the vaccine have just received authorization from the National Health Surveillance Agency (ANVISA) to begin clinical trials.
"There are only some minor adjustments left to be made in the study's protocol before we submit it once again to the approval of the National Council for Research Ethics [CONEP]. We expect to start clinical trials by late October," Ricardo Tostes Gazzinelli, head of the Federal University of Minas Gerais's Vaccine Technology Center (CTV-UFMG), told Agência FAPESP. Gazzinelli is also a senior researcher at Oswaldo Cruz Foundation (Fiocruz), the Ministry of Health's research arm.
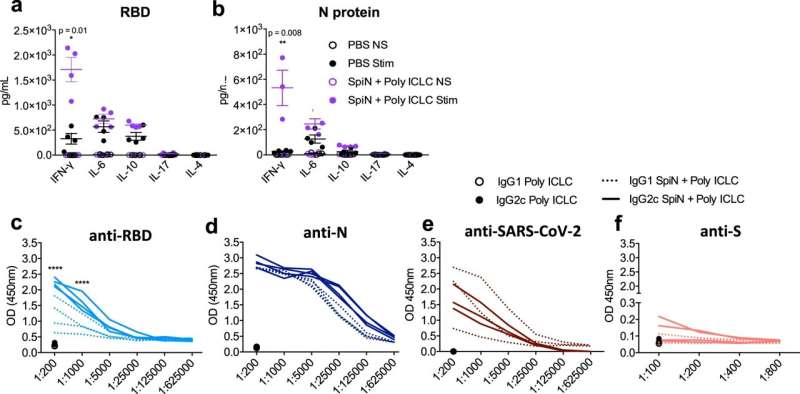
To develop the formulation for the vaccine, the group led by Gazzinelli combined two different SARS-CoV-2 proteins: N (for nucleocapsid, which contains the virus's genetic material) and part of S (for spike, the protein used by the virus to bind to invade human cells). The resulting chimeric molecule is called SpiN. The strategy aims to trigger a cellular immune response consisting of the production of defense cells (T lymphocytes) that specialize in recognizing and killing the novel coronavirus. This type of protection should remain effective even against novel variants.
"The COVID-19 vaccines in use now are designed mainly to trigger the production of neutralizing antibodies against the S protein and prevent the virus from infecting human cells. This is what's known as the humoral immune response. However, with the emergence of variants with many mutations in the S protein, the ability of these antibodies to recognize this antigen has weakened, whereas the N protein is better conserved in the new strains," said Ph.D. candidate Julia Castro, who led the preclinical trials with Gazzinelli supervising.
According to Gazzinelli, who is also a visiting professor at the University of São Paulo's Ribeirão Preto Medical School (FMRP-USP), the vaccine based on the chimeric protein SpiN does not itself trigger the production of neutralizing antibodies, but if given as a booster shot it can stimulate both the humoral immunity produced by prior vaccination and cellular immunity, affording double protection.
Challenge testing
The animal trials were performed at a high-biosafety laboratory at FMRP-USP, thanks to collaboration with João Santana da Silva and Luiz Tadeu Figueiredo, both of whom are also professors there.
The first step was to test the efficacy of the vaccine in mice that had been genetically modified to express ACE2, the human protein to which the virus binds via its spike (S) to infect the cells of the host. This model mimics the severe form of COVID-19.
Some of the mice were given two doses 21 days apart. The others received a placebo. A month later they were exposed intranasally to a high viral load. Different experiments were performed to test the extent to which the vaccine protected them against the wild-type strain of SARS-CoV-2 (isolated in China in 2019), the delta variant (India, 2020) and the omicron variant (South Africa, 2021).
"In the control group, which was given placebo, 100% of the animals infected with the [wild-type] Wuhan strain or delta died. The mice exposed to omicron didn't die but developed a significant pathology in the lungs," Castro said. "In the vaccinated group, all the animals survived infection by all three strains and lung tissue was much more preserved. In addition, viral load was between 50 and 100 times lower."
The next step involved testing the vaccine on a moderate disease model. To do this, the scientists used hamsters, which are naturally infected by the virus but not very effectively. They were given two doses of the vaccine and after a month were exposed to the Wuhan or delta strain. Compared with the control group, the vaccinated hamsters had a viral load that was about ten times lower and fewer signs of lung damage.
Stability and safety
A platform was established at CTV-UFMG to produce the chimeric protein SpiN in genetically modified bacteria. Tests were also performed there to guarantee purity (absence of contaminants in the formulation) and stability (durability at different temperatures).
"The results showed that the vaccine remains viable for two weeks at room temperature and for at least six months when stored at 4 °C," told Gazzinelli, according to whom safety and toxicity tests were performed on rats.
According to Gazzinelli, clinical trials are divided into Phase I and II. Phase I is expected to immunize 80 patients to make sure the vaccine is safe for humans, while Phase II will include a group of 400 volunteers for vaccine safety tests and also for evaluation of the vaccine's immunogenicity—or, in other words, its capacity to induce an effective immune response. Trials will be conducted at UFMG's medical school and will be led by Helton Santiago and Jorge Pinto, both of whom are professors there. They plan to vaccinate people who have already been given any of the available COVID-19 vaccines at least six months previously.
"It will be a booster shot. Volunteers in the control group will receive the AstraZeneca vaccine. We'll then compare levels of neutralizing antibodies to SARS-CoV-2 and T lymphocytes. We expect our formulation to trigger an even stronger cellular immune response," Gazzinelli said.
More information: Castro, J.T. et al, Promotion of neutralizing antibody-independent immunity to wild-type and SARS-CoV-2 variants of concern using an RBD-Nucleocapsid fusion protein, Nature Communications (2022). DOI: 10.1038/s41467-022-32547-y