This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New insights on pertussis vaccines could pave the way for next-gen protection

In a new study published in Cell Host & Microbe, scientists at La Jolla Institute for Immunology (LJI) have compared the immune response of individuals who received older versus newer versions of the whooping cough vaccine. The unexpected findings may help explain the recent rise in whooping cough cases and point to potential targets for the next generation of vaccines.
Whooping cough is a serious and sometimes life-threatening infectious disease caused by the highly contagious bacterium Bordetella pertussis. In 1948, the first whole-cell pertussis (wP) vaccine was made widely available, in which recipients were treated with an inactivated version of the entire bacterium. While extremely effective in producing immunity, the potent vaccine sometimes caused short-term side effects, leading the United States to adopt an acellular pertussis (aP) vaccine in 1996. This vaccine contains only four proteins from B. pertussis.
Clinical cases of whooping cough have risen dramatically in recent decades, and increasingly affect fully vaccinated teenagers and young adults whose ages coincide with the implementation of the aP vaccine. This has led researchers to wonder whether the protection provided by the aP vaccine is somehow less effective than that of the older wP vaccine.
Scientists have characterized human T cell reactivity to the four main antigens present in the aP vaccine, but they haven't had a clear picture of the human immune response to other B. pertussis antigens.
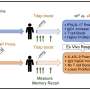
For the new study, scientists in the lab of LJI Professor Alessandro Sette, Dr.Biol.Sci., set out to map the landscape of T cell reactivity against the entire B. pertussis proteome. This closer look at T cell reactivity would then help the researchers determine whether there are differences in the breadth, magnitude, and type of immune response between individuals who received wP versus aP vaccines.
To do this, the researchers acquired blood samples from adults vaccinated with either wP or aP vaccines who had never been diagnosed with whooping cough. Taking advantage of recent technical advances, the scientists were able to characterize the response of participants' T cells to more than 3,000 proteins (ORFs) encoded in the B. pertussis genome.
The researchers anticipated that T cells from individuals treated with the aP vaccine would exhibit responsiveness exclusively to the four B. pertussis antigens present in that vaccine. However, their findings revealed that the overall pattern and magnitude of T cell responses to aP and non-aP vaccine antigens were similar regardless of which vaccine the individual had received.
"We were really surprised to see that," says Sette. "How were these individuals showing immunity to so many antigens not present in the vaccine?"
Sette says the aP vaccine likely produces an immune response to the four main antigens that is effective enough to prevent severe disease, but not very good at preventing subclinical infections. This would lead a large number of people to unknowingly become infected and act as carriers of B. pertussis, spreading the bacteria to those more vulnerable to the disease.
"Our findings suggest that asymptomatic infection, not vaccination, is what drives the T cell response in adults vaccinated with the aP vaccine," says LJI Instructor Ricardo da Silva Antunes, Ph.D., who serves as corresponding author and first author of the study. "The role of asymptomatic infections in the resurgence of whooping cough has likely been underappreciated."
The scientists say that new vaccines that teach the immune system to target an additional set of B. pertussis antigens could be developed to either replace or supplement the current aP vaccine.
The researchers also examined the type of immune response produced against aP and non-aP antigens. Past studies show that childhood aP vaccination tends to produce a type 2 immune response, where T cells produce a specific set of inflammatory molecules to help fight disease. Type 2 responses often block or counteract the more protective type 1 responses.
However, the LJI team found that when T cells from participants who received the aP vaccine were exposed to non-aP vaccine antigens, they did not show any polarization or bias towards type 1 or type 2 responses. Their finding suggests non-aP antigens have the potential to counteract the type 2 bias of aP vaccination and provide a more well-rounded immune response to B. pertussis.
Until these next-generation vaccines can be designed, clinically tested, and distributed, LJI scientists encourage people to take advantage of currently available measures to prevent the spread of whooping cough.
"Because pertussis is so easily transmitted by coughing and close contact, everyone is a potential carrier, so it is paramount that we continue to get vaccinated and boosted in order to protect the infants, children, seniors, and immunocompromised individuals in our communities," says da Silva Antunes.
Additional authors of the study, "T cell reactivity to Bordetella pertussis is highly diverse regardless of childhood vaccination," include Emily Garrigan, Lorenzo G. Quiambao, Sandeep Kumar Dhanda, Daniel Marrama, Luise Westernberg, Eric Wang, Adam Abawi, Aaron Sutherland, Sandra K. Armstrong, Timothy J. Brickman, John Sidney, April Frazier, Tod J. Merkel, and Bjoern Peters.
More information: Ricardo da Silva Antunes et al, T cell reactivity to Bordetella pertussis is highly diverse regardless of childhood vaccination, Cell Host & Microbe (2023). DOI: 10.1016/j.chom.2023.06.015