This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Lung cancer cells covertly thrive in brain under guise of protection, study finds
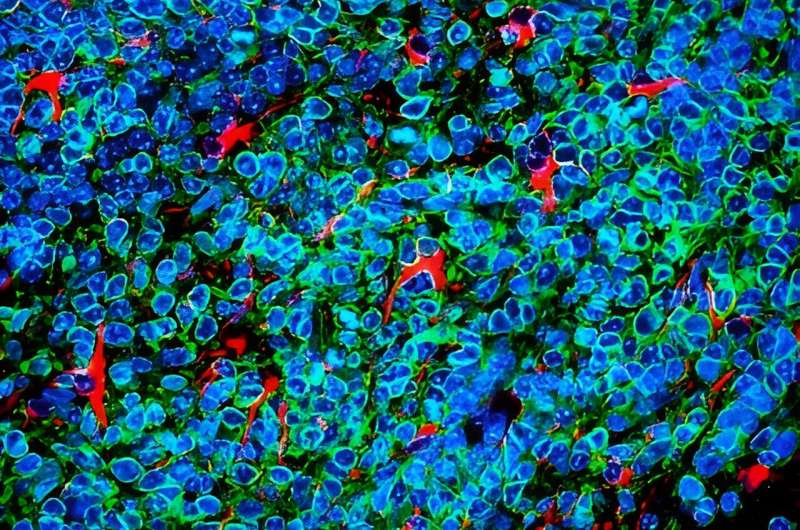
Lung cancer cells that metastasize to the brain survive by convincing brain cells called astrocytes that they are baby neurons in need of protection, according to a study by researchers at Stanford Medicine.
The cancer cells carry out their subterfuge by secreting a chemical signal prevalent in the developing human brain, the researchers found. This signal draws astrocytes to the tumor and encourages them to secrete other factors that promote the cancer cells' survival. Blocking that signal may be one way to slow or stop the growth of brain metastases of small cell lung cancer, which account for about 10% to 15% of all lung cancers, the researchers believe.
Astrocytes play a critical role in maintaining nerve function and connectivity in the adult brain. They are also important during brain development, when they facilitate connections between developing neurons.
The researchers studied laboratory mice, human tissue samples and human mini-brains, or organoids, grown in a lab dish to dissect the unique relationship between the cancer cells and their "big sister" astrocytes, which hover nearby and shower them with protective factors.
"Small cell lung cancers are known for their ability to metastasize to the brain and thrive in an environment that is not normally conducive to tumor growth," said professor of pediatrics and of genetics Julien Sage, Ph.D. "Our study suggests that these cancer cells reprogram the brain microenvironment by recruiting astrocytes for their protection."
Sage, the Elaine and John Chambers Endowed Professor in Pediatric Cancer, is the senior author of the study, which was published online Oct. 2 in Nature Cell Biology. Postdoctoral scholar Fangfei Qu, Ph.D., is the lead author of the study.
Invasion of the brain
Small cell lung cancer is particularly talented at metastasizing to the brain. So talented, in fact, that about 15% to 20% of people already have clusters of cancer cells in their brains when their lung tumors are first diagnosed. As the cancer progresses, about 40% to 50% of patients will develop brain metastases. The problem is so prevalent, and the clinical outcome so dire, that clinicians recommend cranial radiation even before brain metastases have been found.
It's been tough to study how and why small cell lung cancer has such an affinity for the brain, however. Brain metastases are rarely biopsied or removed because doing so hasn't been shown to affect a patient's survival, and brain surgery is so invasive. Additionally, laboratory mice with small cell lung cancers rarely develop metastases in the brain, perhaps due to subtle biological differences between species.
Small cell lung cancers have another distinguishing feature. They are what's known as neuroendocrine cancers, meaning they arise from cells with similarities to both neurons and hormone-producing cells. Neuroendocrine cells link the nervous system with the endocrine system throughout the body, including in the lung.
Sage and his colleagues wondered whether neuronal-associated proteins on the surface of small cell lung cancer cells give them a leg up when the cells first begin to infiltrate the brain.
"We know the brain is full of neurons," Sage said. "Maybe that's why these cancer cells with some neuronal traits are happy in the brain and are accepted into that environment."
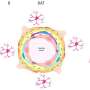
Qu and Sage developed a way to inject mouse small cell lung cancer cells grown in the laboratory into the brains of mice to spark the development of brain tumors. They saw that astrocytes, which belong to a family of cells called glial cells, flocked to the infant tumors and began to churn out proteins critical during brain development, including factors that stimulate nerve growth.
A plethora of astrocytes
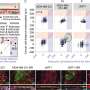
A similar siren call happens in human brains, they noted: Brain tissue samples from people who had died of metastatic small cell lung cancer, shared by professor of pathology and paper co-author Christina Kong, MD, had many more protective astrocytes in the interior of the tumors than did metastases of melanoma, breast cancer and another type of lung cancer called adenocarcinoma.
Qu worked with assistant professor of pediatrics and co-author Anca Pasca, MD, to fuse aggregates of small cell lung cancer, lung adenocarcinoma or breast cancer cells with what are called cortical organoids—clumps of brain cells including neurons and astrocytes grown in a laboratory dish that begin to mimic the organization and connectivity of a human cortex. Within 10 days, many more protective astrocytes had infiltrated the small cell lung cancer pseudo-tumors than the adenocarcinoma or breast cancer.
"This showed us that the astrocytes actively move toward the small cell lung cancer cells, rather than simply being engulfed by the growing tumor," Sage said. "What's more exciting, though, is that these organoids, or mini-brains, realistically model the developing human brain. So, we're no longer relying on a mouse model. It's a perfect system to study brain metastases."
Further research showed that the small cell lung cancer cells summon protective astrocytes by secreting a protein called Reelin that mediates the migration of neuronal and glial cells during brain development. Triggering Reelin expression in mouse breast cancer cells injected into the brain significantly increased the number of astrocytes in the resulting tumors in the mice, and the tumors were larger than in control animals injected with cells with low Reelin expression.
The apparent reliance of the cancer cells on chemical signals and responses specific to the developing brain may give clues for the development of future therapies, Sage believes.
"Some of these signals may not be as relevant or as highly expressed in the adult brain," Sage said. "As a result, perhaps they could still be targeted to slow or prevent brain metastases without harming a normal brain. This might be an important window of opportunity for therapy."
More information: Fangfei Qu et al, Crosstalk between small-cell lung cancer cells and astrocytes mimics brain development to promote brain metastasis, Nature Cell Biology (2023). DOI: 10.1038/s41556-023-01241-6