This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
New tool reveals how drugs affect men, women differently, and could lead to safer medications
UVA Health researchers have developed a powerful new tool to understand how medications affect men and women differently, and that will help lead to safer, more effective drugs in the future.
Women are known to suffer a disproportionate number of liver problems from medications. At the same time, they are typically underrepresented in drug testing. To address this, the UVA scientists have developed sophisticated computer simulations of male and female livers and used them to reveal sex-specific differences in how the tissues are affected by drugs.
The new model has already provided unprecedented insights into the biological processes that take place in the liver—the organ responsible for detoxifying the body—in both men and women. But the model also represents a powerful new tool for drug development, helping ensure that new medications will not cause harmful side effects.
"There are incredibly complex networks of genes and proteins that control how cells respond to drugs," said UVA researcher Jason Papin, Ph.D., one of the model's creators. "We knew that a computer model would be required to try to answer these important clinical questions, and we're hopeful these models will continue to provide insights that can improve health care."
Harmful effects from drugs
Papin, of UVA's Department of Biomedical Engineering, developed the model in collaboration with Connor Moore, a Ph.D. student, and Christopher Holstege, MD, a UVA emergency medicine physician and director of UVA Health's Blue Ridge Poison Center. "It is exceedingly important that both men and women receive the appropriate dose of recommended medications," Holstege noted. "Drug therapy is complex and toxicity can occur with subtle changes in dose for specific individuals."
Before developing their model, the researchers first looked at the federal Food and Drug Administration's Adverse Event Reporing System to evaluate the frequency of reported liver problems in men and women. The scientists found that women consistently reported liver-related adverse events more often than men.
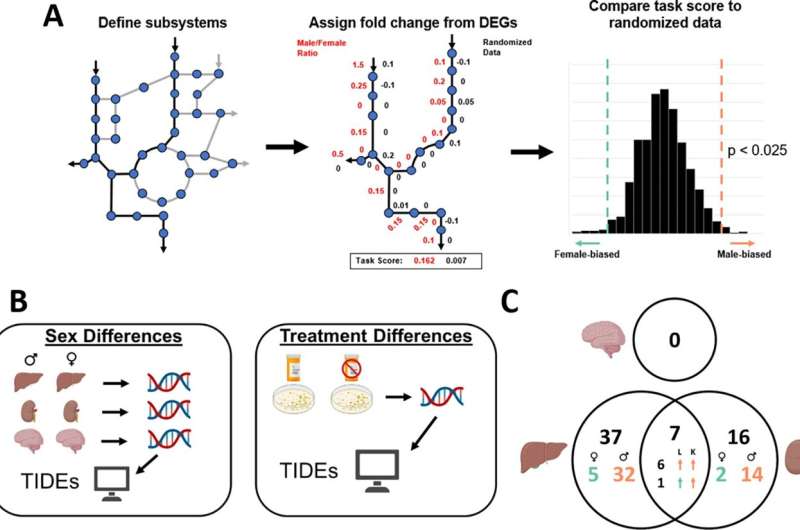
The researchers then sought to explain why this might be the case. To do that, they developed computer models of the male and female livers that integrated vast amounts of data on gene activity and metabolic processes within cells. These cutting-edge liver simulations provided important insights into how drugs affect the tissue differently in men and women and allowed the researchers to understand why.
"We were surprised how many differences we found, especially in very diverse biochemical pathways," said Moore, a biomedical engineering student in Papin's lab. "We hope our results emphasize how important it is for future scientists to consider how both men and women are affected by their research."
The work has already identified a key series of cellular processes that explain sex differences in liver damage, and the scientists are calling for more investigation of it to better understand "hepatotoxicity"—liver toxicity. Ultimately, they hope their model will prove widely useful in developing safer drugs.
"We're hopeful these approaches will be help address many other questions where men and women have differences in drug responses or disease processes," Papin said. "Our ability to build predictive computer models of complex systems in biology, like those in this study, is truly opening all kinds of new avenues for tackling some of the most challenging biomedical problems."
The researchers have published their findings in PLOS Computational Biology.
More information: Connor J. Moore et al, Metabolic modeling of sex-specific liver tissue suggests mechanism of differences in toxicological responses, PLOS Computational Biology (2023). DOI: 10.1371/journal.pcbi.1010927