This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researchers reveal uncharted liver-focused pathway in gene therapy immune responses
Indiana University School of Medicine researchers have uncovered vital insights regarding a liver trigger that blocks an undesired immune response from gene therapy, surprisingly resulting in activating specific immune cells despite the liver's typical role in suppressing immune responses.
The findings, published in Molecular Therapy, may pave the way for change in immunomodulation strategies for desired and long-lasting effects of gene therapy.
Gene therapy treatments involve replacing or introducing a healthy copy of the flawed gene that's causing the genetic disease. The process is often accomplished through a viral vector—in this case, a small, harmless virus called adeno-associated virus (AAV), which carries the healthy gene to deliver it to the target organ. Unfortunately, a notable challenge in conventional gene therapy approaches lies in the immune system's tendency to reject certain treatments.
"Occasionally, gene therapy can prompt an adverse immune reaction when the body mistakenly perceives the treatment as a viral threat," said Roland Herzog, Ph.D., Riley Children's Foundation Professor of Immunology at IU School of Medicine and the study's lead author. "Our goal is to explore innovative approaches in gene therapy, striving for methods with higher success rates to benefit patients."
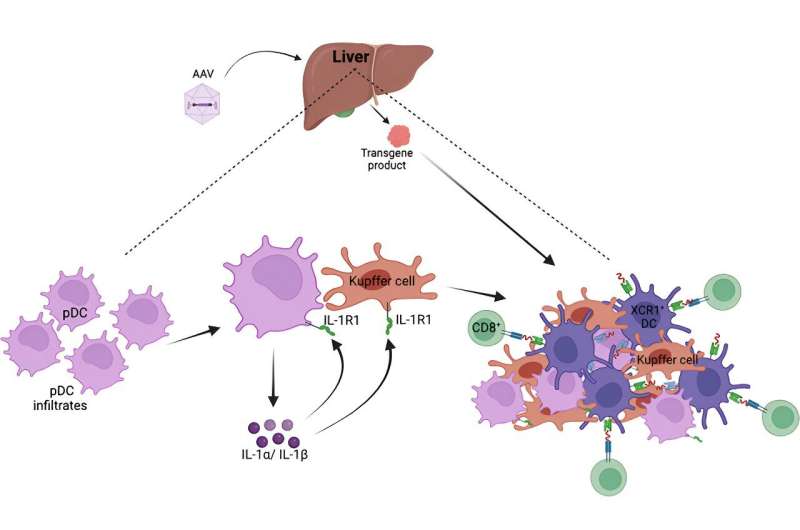
The latest research, led by Herzog and Sandeep Kumar, Ph.D. of the IU School of Medicine's Herman B Wells Center for Pediatric Research, found that certain immune cells, called cytotoxic T lymphocytes, can sometimes be triggered in the liver despite the organ's immune suppressive properties.
Surprisingly, and different from what was known about these immune mechanisms in response to AAV gene therapy, Kumar found that the receptor for a cytokine called interleukin-1 was critical for triggering the response that may result in immune rejection. Importantly, the team was able to prevent the immune response by blocking this pathway. These findings signify an important step to ensure more targeted and controlled immune responses.
"Our data showed that though treatments targeting the liver provide a roadmap for safer and more precise gene therapy options, additional immunomodulation strategies might be needed for long-lasting therapeutic benefit," said Kumar, the study's first author and an assistant research professor of pediatrics at IU School of Medicine.
The study's implications extend beyond liver gene therapy, as the group's findings offer a promising direction for future clinical applications that may benefit several genetic diseases.
"The concept of preserving gene therapies by blocking certain immune responses can be applied to many genetic diseases like hemophilia, lysosomal storage disorders, and metabolic diseases," Herzog said. "This breakthrough is incredibly promising for the field, and we eagerly anticipate its broad adaptation in clinical trials, medications, and gene therapy protocols."
More information: Sandeep R.P. Kumar et al, TLR9-independent CD8+ T Cell Responses in Hepatic AAV Gene Transfer Through IL-1R1-MyD88 Signaling, Molecular Therapy (2023). DOI: 10.1016/j.ymthe.2023.11.029