This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Promising new therapeutic approach for treating metastatic pancreatic cancer

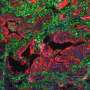
A research paper published in Nature Cancer details new insights into the role of efferocytosis—the burying of dead cells—in pancreatic cancer that spreads to the liver.
Liver metastasis occurs in 40–50% of people with pancreatic ductal adenosarcoma (PDAC), and there are currently no effective therapies to cure pancreatic cancer patients that have liver metastasis.
Led by University of Liverpool's Professor Michael Schmid and colleagues, this study found PDAC metastases to show high levels of immunosuppressive macrophages, a type of white blood cell that promotes tumor growth.
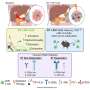
The researchers discovered that blocking the efferocytosis pathway during early-stage metastasis prevented this immunosuppressive activity in macrophages, restoring T cell activation and reducing metastatic tumor burden.
Lead author of the study, Professor Michael Schmid, said, "In pancreatic cancer, malignant tumor cells often spread to the liver. Our data show that the generation of a supportive metastatic 'niche' in the liver is critical for the effective outgrowth of malignant cells at the distant site.
"Our findings suggest that a particular type of immune cells orchestrate the formation of the metastatic niche by reprogramming other immune cells, thereby creating an immunosuppressed metastatic microenvironment, where malignant cells are able to hide from an anti-tumor immune response. Targeting this particular type of innate immune cells or interfering with their immunosuppressive functions could serve as a promising therapeutic approach for patients with metastatic pancreatic cancer."
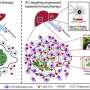
First author Dr. Yuliana Astuti added, "Using single cell technologies, we found an underappreciated diversity of macrophages in pancreatic cancer liver metastases. We identified that in the liver, metastasis-associated macrophages with opposite functions coexist, some exhibiting immunostimulatory and others immunosuppressive features.
"Interestingly, further temporal analysis revealed that liver metastases is accompanied by increased liver tissue cell death and that the engulfment of dead cells acts as a key driver to reprogram macrophages towards an immunosuppressive phenotype. Our study provides proof-of-principle that tailored targeting of specific macrophages restores tumor immunity and inhibits PDAC metastasis."
Key contributor Professor Ainhoa Mielgo commented, "Pancreatic cancer is a very aggressive cancer type that often spreads to the liver. We currently have no effective therapies to cure pancreatic cancer patients that have liver metastasis. These findings are really exciting because they reveal a targetable mechanism by which pancreatic cancer cells spread and grow in the liver. Our hope and goal now is to translate these lab discovery into the benefit of patients."
"This study is a product of a fantastic collaborative effort of scientists, medical oncologists, surgeons and patients working together to find better treatments for pancreatic cancer patients," Professor Schmid added.
Led by the University of Liverpool, the study also involved researchers from Cancer Research-UK Scotland Institute, the University of Glasgow and the University of Edinburgh.
More information: Yuliana Astuti et al, Efferocytosis reprograms the tumor microenvironment to promote pancreatic cancer liver metastasis, Nature Cancer (2024). doi.org/10.1038/s43018-024-00731-2 , www.nature.com/articles/s43018-024-00731-2