This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists reveal how a potassium ion channel reprograms energy production in cancer cells
Researchers have revealed how a channel that controls cellular potassium levels causes metabolic rewiring in breast cancer cells, promoting tumor growth.
The study, published in eLife, was described by the editors as providing convincing evidence that an intracellular subpopulation of a specific potassium channel reprograms breast cancer cells towards the Warburg phenotype, one of the metabolic hallmarks of cancer.
Ion channels are gateways within cells that tightly control the inward and outward flow of essential ions, such as potassium and calcium, and are thought to be critically linked to cancer malignancy and progression.
Cancer cells show increased energy demands due to their high growth rates and can switch their metabolism from a process requiring oxygen to one that allows them to grow in oxygen and nutrient-poor environments—known as the Warburg effect.
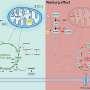
Researchers at the Lukowski lab, University of Tübingen, Germany, together with inter-/national collaboration partners, have now demonstrated that these metabolic processes rely on ions such as potassium and calcium, and their dynamics are regulated by the "large-conductance Ca2+ activated K+ channel"—or BKCa. This channel has been previously reported to promote breast cancer progression, but the exact mechanism underlying this effect was unknown.
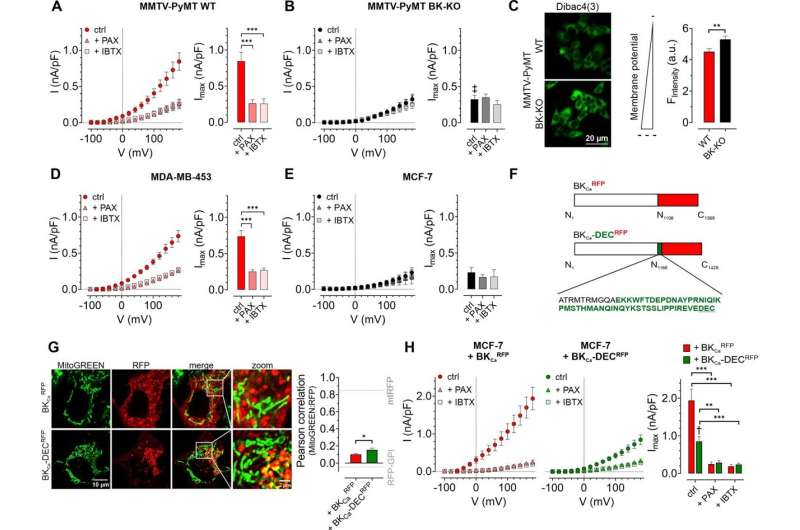
To close this gap in knowledge, the multidisciplinary research team used BKCa-proficient and deficient human cancer cell lines, as well as cancer cells from mice lacking BKCa to study the role of different BKCa subpopulations in cancer cell metabolism and growth. They found that breast cancer cells lacking BKCa had lower intracellular calcium levels. Moreover, this change in calcium levels occurred not only in the main body of the cell but also within substructures such as mitochondria—the tiny power plants that cells use to produce energy.
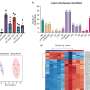
Given these results, the team explored whether BKCa also affected metabolic processes in breast cancer cells. As they anticipated, in cells with BKCa there was an increased output of lactate and lower oxygen consumption than in breast cancer cells without BKCa, suggesting a metabolic rewiring mediated by this type of ion channel.
To explore this further, the authors then looked at how different sources of energy are used in the mitochondria when BKCa is functional. Cells can use and generate energy in the form of a molecule called ATP. When BKCa is present in mitochondria, cells prefer to consume rather than generate ATP. Moreover, this switch causes increased hydrogen peroxide levels which, in turn, cause further genetic damage and contributes to cancer progression.
Having found that BKCa helps cancer cells with reprogramming their metabolism, Dr. Bischof, first author of the study, specifically looked for evidence of the Warburg effect by measuring lactate concentrations over time; higher levels of lactate secretion imply a shift towards metabolism that does not use oxygen.
As expected, the presence of BKCa not only increased lactate secretion, but also increased the ability of breast cancer cells to grow without oxygen. Together, these results point to a significant malignancy-promoting effect of mitochondrial BKCa in breast cancer.
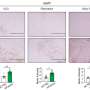
Finally, Bischof et al. explored whether mitochondrial BKCa was present in primary human-derived breast cancer tissue. They found that 551 tissue samples tested positively, with 10 showing significantly high expression of BKCa.
"Our experiments highlight the presence of an ion channel within the energy production machinery in breast cancer cells that promotes profound bioenergetic changes and ultimately triggers the growth of breast cancer cells in an oxygen-depleted environment, such as that found in a solid tumor," concludes senior author Robert Lukowski, Professor of Experimental Pharmacology at the University of Tübingen.
"Taken together, expression of mitochondrial BKCa could be used as a prognostic or therapeutic marker in breast cancer patients and presents a potentially new anti-cancer treatment strategy."
More information: Helmut Bischof et al, mitoBKCa is functionally expressed in murine and human breast cancer cells and potentially contributes to metabolic reprogramming, eLife (2024). DOI: 10.7554/eLife.92511.3