Genome instability studies could change treatment for cancer and other diseases
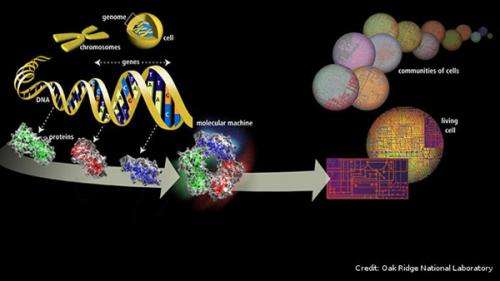
Counterintuitive as it may seem, genetic mutation is key to our evolution and survival.
As our cells grow, reproduce, and die, DNA is repeatedly replicated and repaired, and bits and pieces of its sequences are perpetually changed, misplaced, and swapped in the process, thus producing mutations. These mutations create genetic variation, which results in different observable traits or phenotypes—providing material for the process of natural selection to act upon and driving the evolution of fitter populations.
But mutations aren't always beneficial for the individual; in some cases, they can be harmful, leading to inherited or somatic genetic diseases. A growing number of neurodegenerative and neuromuscular inherited diseases are being linked to mutations that arise from instability in one particular group of DNA sequences called microsatellites. And geneticists have also observed microsatellite instability in the genomes of a broad variety of cancer cells, including those found in colon, rectal, endometrial, ovarian, lung, melanoma, pancreatic, gastric, and bladder cancers.
"Microsatellites, which are repetitive DNA sequences with high rates of instability and mutation, constitute roughly 3-4 percent of the human genome and are one of many types of repeats in the genome," says Kateryna Makova, a professor of biology at Penn State and director of the Center for Medical Genomics.
"The instability inherent in microsatellites is a problem," Makova says, "because it frequently leads to genetic diseases. It can lead to cancer, for example, and it can lead to other hereditary diseases, but it is also one of the evolutionary mechanisms to create novelty in the genome and thus a substrate for natural selection to operate on. So it can be viewed as disadvantageous from one standpoint, but also highly advantageous from another standpoint."
Research at the Center for Medical Genomics
Six years ago, Makova and colleagues Kristin Eckert and Francesca Chiaromonte—all Huck Institutes affiliates in the Center for Medical Genomics—began employing a unique collaborative approach, combining computational biology, statistical modeling, and wet-bench experiments, to elucidate microsatellites' mutational behavior and the genomic events defining their evolution.
"Over the past 12 years, our laboratory has extensively studied the mechanisms of microsatellite mutation in human cells, using both cell lines and purified DNA polymerases," says Eckert, a professor of pathology and of biochemistry and molecular biology at the Penn State College of Medicine in Hershey.
"Our direct experimental analyses," Eckert says, "demonstrated that the mutability of each microsatellite is dependent primarily upon features intrinsic to the repeated DNA itself (repeat size, length, and sequence), which is the same conclusion regarding microsatellite mutability that was reached by the Makova and Chiaromonte groups, who fit statistical models to microsatellite mutability data inferred from primate genome comparisons.
"The fact that the same mechanisms that appear to underlie mutations generated in a test tube also appear to underlie the mutations generated during primate genome evolution was very remarkable, and encouraged us to embark on a collaborative research project."
Between 2008 and 2012, with important contributions from graduate students Yogeshwar Kelkar and Akarachai Fungtammasan, the researchers published several key papers identifying fundamental properties of microsatellites. Collectively, these papers established genomic "signatures" characterizing these sites and the genomic landscape that surrounds them, how they arise and die in the genome, and the factors that influence their instability and rates of mutation.
In 2013, with significant contributions from graduate students Guruprasad Ananda, Beverly Baptiste, and Erin Walsh, the researchers have published a pair of papers further detailing the mutational behavior of microsatellites and their underlying mechanisms.
How small is micro?
An article earlier this year in Genome Biology and Evolution (GBE) represents the first comprehensive genome-wide analysis of the mutational behavior of tandem repeats—the broader category of repetitive DNA sequences to which microsatellites belong.
"With the GBE paper, our major question was 'What is a microsatellite?'" says Makova. "We knew that it is a repetitive DNA sequence. But with just, say, two repeats—is it already a microsatellite?"
"In other words," says Chiaromonte, "is there a typical length at which these sequences acquire the mutational behavior of microsatellites? This is a very important and longstanding question in the field of genomics."
"Using data from the 1,000 Genomes Project," says Makova, "we defined the minimum number of repeats for a sequence to be considered a microsatellite—when the mutation rates become very high and instability becomes high, as well."
"In parallel," says Eckert, "an in vitro assay was developed by our lab to determine mutational mechanisms of short tandem repeat sequences as a function of repeat length. In both studies, a critical threshold length was identified wherein tandem repeats change their mutational behavior and become microsatellites."
"This discovery has implications for medical genomics in studies of chromosomal fragile sites," says Makova, "and also for molecular ecology, since scientists use these same loci—microsatellites—to study diversity within populations."
Predicting mutations
Building on the research presented in GBE, an article in Genes, Genomes, Genetics (G3) is focused on mutational mechanisms in mature microsatellites, which are long microsatellites that undergo rapid mutations.
Prior evolutionary studies have indicated that long microsatellites are biased towards a type of mutation known as repeat expansion, which increases the number of times that a DNA sequence is repeated and which is known to cause inherited disorders such as Huntington's disease, myotonic dystrophy, fragile X syndrome type A, Friedreich's ataxia, and a number of spinocerebellar ataxias.
"In the G3 paper, our collaborative group provided a biochemical mechanism to explain the directionality bias favoring expansions in human cells," says Eckert, "and we showed computationally that mature microsatellites are highly abundant within protein coding regions of the human genome. Together with Maria Krasilnikova, our group also uncovered mechanistic parallels with the rare trinucleotide microsatellite expansions that are involved in neurological diseases."
"Our research may have direct relevance to public health and personalized medicine," Eckert notes. "For example, the new information gained by our research can be used to predict the probability of each microsatellite within the genome to undergo mutation. This will have major importance for assessing an individual's disease risks, especially in the era of whole genome sequencing."
Future directions
Ultimately, the body of knowledge that these researchers are working to build can be used to inform any number of scientific fields relying upon genetics, genomics, and evolution—even forensics, which employs microsatellites as genetic markers for DNA fingerprinting.
But the greatest impact may still be in medical genomics—in the development of personalized medicine and therapeutic strategies to treat and reduce the impact of inherited diseases that stem from instability in the human genome.
"We are currently working on a very interesting project on interrupted microsatellites," says Makova, "which are loci where a microsatellite sequence is interrupted by the insertion of a nucleotide that is not part of the sequence. This has been shown in some cases to greatly stabilize the microsatellite, because it reduces the efficiency of strand slippage, which is the major mechanism driving mutation. It's already been shown that these interruptions can stabilize microsatellites to the point where the incidence of disease is decreased. What we're seeing now is that, in fact, this effect is global—we suspect that it is happening across the genome.
"In the future, this idea could potentially be used to treat cancers and other diseases by developing gene therapies that work by creating stabilizing interruptions in long, highly mutable microsatellite sequences."
More information: g3journal.org/content/3/3/451