Researchers identify proteins that may help brain tumors spread
Scientists at the University of Alabama at Birmingham have identified a molecular pathway that seems to contribute to the ability of malignant glioma cells in a brain tumor to spread and invade previously healthy brain tissue. Researchers said the findings, published Sept. 19, 2013, in the journal PLOS ONE, provide new drug-discovery targets to rein in the ability of these cells to move.
Gliomas account for about a third of brain tumors, and survival rates are poor; only about half of the 10,000 Americans diagnosed with malignant glioma survive the first year, and only about one quarter survive for two years.
"Malignant gliomas are notorious, not only because of their resistance to conventional chemotherapy and radiation therapy, but also for their ability to invade the surrounding brain, thus causing neurological impairment and death," said Hassan Fathallah-Shaykh, M.D., Ph.D., associate professor in the UAB Department of Neurology. "Brain invasion, a hallmark of gliomas, also helps glioma cells evade therapeutic strategies."
Fathallah-Shaykh said there is a great deal of interest among scientists in the idea that a low-oxygen environment induces glioma cells to react with aggressive movement, migration and brain invasion. A relatively new cancer strategy to shrink tumors is to cut off the tumor's blood supply – and thus its oxygen source – through the use of anti-angiogenesis drugs. Angiogenesis is the process of making new blood vessels.
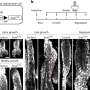
"Stop angiogenesis and you shut off a tumor's blood and oxygen supply, denying it the components it needs to grow," said Fathallah-Shaykh. "Drugs that stop angiogenesis are believed to create a kind of killing field. This study identified four glioma cell lines that dramatically increased their motility when subjected to a low-oxygen environment – in effect escaping the killing field to create a new colony elsewhere in the brain."
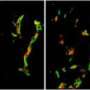
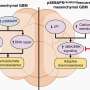
Fathallah-Shaykh and his team then identified two proteins that form a pathway linking low oxygen, or hypoxia, to increased motility.
"We identified a signaling protein that is activated by hypoxia called Src," said Fathallah-Shaykh. "We also identified a downstream protein called neural Wiskott-Aldrich syndrome protein (N-WASP), which is regulated by Src in the cell lines with increased motility."
The researchers then used protein inhibitors to shut off Src and N-WASP. When either protein was inhibited, low oxygen lost its ability to augment cell movement.
"These findings indicate that Src, N-WASP and the linkage between them – which is something we don't fully understand yet – are key targets for drugs that would interfere with the ability of a cell to move." said Fathallah-Shaykh. "If we can stop them from moving, then techniques such as anti-angiogenesis should be much more effective. Anti-motility drugs could be a key component in treating gliomas in the years to come."
More information: www.plosone.org/article/info%3Adoi%2F10.1371%2Fjournal.pone.0075436