May 23, 2013 report
Controlling mood through the motions of mitochondria
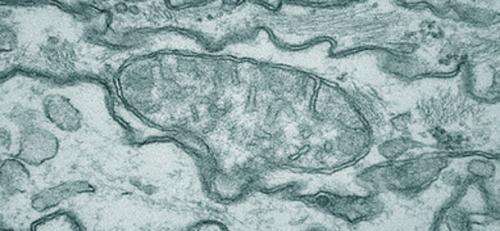
(Medical Xpress)—Regulating the distribution of power in neurons is done by a system that makes the national electric grid look simple by comparison. Each neuron has several thousand mitochondria confined into narrow neuritic farms. Near-incessant motion within these channels forces mitochondria to transact constant fusion and fission events whose roles in genetic repair are just beginning to be understood. Many common neurodegenerative diseases have been found to have an underlying mitochondrial deficit either in their ability to produce energy, or deal with the oxidative byproducts of producing that energy. It has recently emerged that deficits in the ability of mitochondria to translocate, fuse, and divide also contribute significantly to disease. A new study in Plos One now suggests that alterations in the way mitochondria move may directly underlie even more esoteric phenomena—like mood.
The authors of the new study had previously reported on the various effects of the antipsychotics like lithium, valproate and paliperidone at the synaptic level. Noting that mitochondrial defects have been implicated in schizophrenia and bipolar disorder, they made the leap of faith postulate that mood stabilizers might act by altering the rate and direction of mitochondrial movements. To support these claims they investigated the effects of these drugs on what they call the phosphoproteome—the sum total, and states, of all the phosphorylated proteins in the cell.
In the familiar models of transport, the cytoskeleton provides the track upon which molecular motors push or pull mitochondria and other organelles. Larger range migrations are done on microtubular structures, while shorter range movements are facilitated by actin. The activity of this infrastructure is regulated by its phosphorylation state, and also that of the many accessory proteins involved in stabilization and attachment.
The authors did an extensive analysis of protein phosphorylation was using a procedure that isolated only the synaptic junctions of neurons. They also tracked mitochondrial movements in slice preparations from the cortex, and in cultured cells. They found that mood stabilizer drugs promoted the migration of mitochondria into synapses, presumably to ensure sufficient energy for neurotransmission.
Other studies have shown that synapses which support sustained firing of spikes at high rates have uniquely sized and shaped mitochondria. In the Lamprey, for example, smaller mitochondria are closely associated with release sites. Prototypical synapses with narrow spine necks may not possess an associated mitochondria at all times. In fact this is still a much debated question in neuroscience. Construction of linked chains of mitochondrial synticium, through the regulation fission and fusion, is perhaps one way to stabilize morphology in size-restricted and metabolically demanding locations.
Based on previous studies, the authors also suggest that direction of mitochondrial motion is a key factor. In axons where microtubules are consistently arranged with their "plus" ends distal, mitochondria with intact membrane potentials tend to migrate in an anterograde fashion. When they are more depolarized, they travel in a retrograde fashion back to the cell body. Presumably, defective mitochondria returning to the cell would either fuse with lysosomes for degradation, or fuse with other outbound mitochondria to freshen up miscoded DNA or transcript. In this way, mitochondria would essentially provide a shuttle for genetic material to reach the full extent of the axon. Incidentally, this concept calls to mind the way ants communicate information between the nest and the periphery using local "back-and-worth" chemosensory exchanges which appear to us only as crude head knocks.
Other drugs, like Clozapine apparently cause mitochondria to balloon and congregate around the nuclei. Haliperidol also has been reported to have related detrimental effects. The idea that the effects of these kinds of drugs extends beyond simplistic concepts of receptor binding kinetics, to more basic structural effects that can be readily observed has gained popularity. For example, independent freelance scientist Ethan Perlstein studies the effects of various psychoactive drugs in yeast. While yeast do not have easily measured mood swings, the intrinsic effects of various drugs on cell machinery can be studied, at least at the discovery stage, with much less effort.
In watching video footage of mitochondrial motions it is perhaps difficult to attribute the full dynamic behavior of mitochondria to a few comparatively miniscule molecular motors, particularly motion within a fairly constrained skeletal meshwork. If mitochondria have other tricks to move about the cell, perhaps riding action potential waves, seeding cytoskeletal propulsion like Listeria bacteria, or otherwise directing asymmetric ion pumping or flow across their membranes, they, rather then their neuron hosts, might be seen as the prime movers in the brain.
Much of the presumed action of various drugs on mitochondria is through neurotransmitter systems, in particular serotonin and dopamine. These signals ultimately cause calcium influx on both sides of the synapse, and subsequently mitochondria are scuttled to assist in the mopping and redistribution. While attributing mood to a chemical has been long accepted, it may be a while before people accept the idea that changing mitochondrial movement statistics might better captures the effect.
Mitochondria source more than just ATP and calcium to various parts of the cell. As new probes like voltage and calcium imaging, and established techniques like Patch clamping are applied to mitochondria, a finer-grained picture of their correlations to the larger spiking activity of neurons may emerge. The critical tool for observing mitochondria may ultimately be high resolution temperature imaging. Might mitochondria move along temperature gradients inside a neuron. If so, in what direction?
More information: Corena-McLeod M, Walss-Bass C, Oliveros A, Gordillo Villegas A, Ceballos C, et al. (2013) New Model of Action for Mood Stabilizers: Phosphoproteome from Rat Pre-Frontal Cortex Synaptoneurosomal Preparations. PLoS ONE 8(5): e52147. doi:10.1371/journal.pone.0052147
© 2013 Medical Xpress