Sweat glands heal injuries
Our body's sweat glands are a source of stem cells particularly suited to healing wounds – stem cells that form new skin cells and manage the healing process. They are not rejected by the body and can be obtained without a hospital stay.
It all began with the pancreas. Prof. Charli Kruse, head of the Fraunhofer Research Institution for Marine Biotechnology EMB in Lübeck, still remembers it well. The researchers had isolated some cells taken from the organ in a petri dish as part of their research – their aim was to investigate the function of the protein Vigilin, which is produced in the gland cells. "Suddenly we realized that these cells were reproducing in an unusual way, since the microscope showed not only gland cells in the dish but nerve and muscle cells, too." Stem cells had formed out of the gland tissue and multiplied to form a variety of different cells. It quickly became apparent that other gland cells shared the same capability: "We slowly worked our way outward from the internal organs until we got to the skin – and the sweat glands. Again, this yielded the same result: a petri dish full of stem cells." Up to now the sweat glands haven't really received much attention since laboratory animals such as mice or rats have them only on their paws. A human being, on the other hand, possesses up to three million – predominantly on the soles of the feet, palms of the hand, armpits and forehead.
Healing stem cells obtained from the armpit
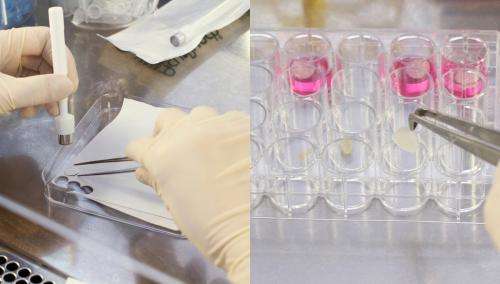
Biologists and medics use stem cells to obtain new tissue to replace cells that are damaged or diseased. They play a particularly important role in healing wounds. Stem cells taken from the patient's own body are ideal because there is no chance of the body rejecting them. Getting at them, however, requires a cumbersome operation to extract the stem cells either from bone marrow or from the blood. "The sweat glands are significantly easier to get to. All that is required is a short walk-in walk-out visit to your dermatologist. We can obtain stem cells from less than 3 millimeters of underarm skin," explains Kruse. When grafted to a skin injury, these stem cells can have a very beneficial effect on the healing process. Whether it is the cells themselves that build new skin cells and blood vessels or whether their role is to manage healing processes by releasing growth hormones that in turn activate immune cells is currently undergoing investigation.
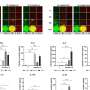
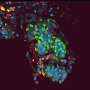
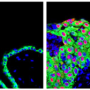
Tests conducted by the scientists on animals and on human skin in the petri dish have demonstrated the beneficial effect of stem cells in healing injuries. In these tests, the researchers took a skin sample and separated it under a microscope into millimeter-sized living sweat glands. They then multiplied the cells contained in these glands outside the body, and in doing so induced them to form cells of different types: "We used these to populate a substrate, and then put the substrate on a wound we had previously generated on a skin specimen." The findings showed that the wound healed significantly faster and better with the stem cells than without them. The substrate gives the cells a solid structure and can be made of something such as collagen, a structural protein of connective tissues in the human body, which can later be replaced by the body's own fibrous proteins. "Without this structure the cells would be taken by the bloodstream and transported away from the site. It is imperative that the cells stay as fixed as possible on the site of the wound as only then can they react with the skin and take part in the healing process," says Kruse. He works closely on wound repair with the department for plastic surgery at the University of Lübeck.
Since the end of last year, Fraunhofer EMB has been collaborating with Bioenergy CellTec GmbH, which has moved from Cologne to Lübeck. To develop new products for wound repair this biotechnology company makes use of a novel substrate – a biopolymer particularly suited to combination with cells. It is hydrophilic – meaning it attracts and incorporates water – and treated in a way that makes it a particularly attractive place for cells to colonize. Now both partners want to pool their developments and team together to produce products that will heal wounds faster and better. "Particularly in the case of chronic wounds, which often do not heal even over a long period of time, there is still no effective course of treatment," says Dr. Kathrin Adlkofer, managing director of Bioenergy. These permanently open wounds are caused by diseased veins or arteries, diabetes, tumors, infections or skin diseases.
The Lübeck scientists already have further applications in mind: "Not only are stem cells from sweat glands easy to cultivate, they are extremely versatile, too." Kruse and his team are already testing a treatment for macular degeneration – a disease of the retina that affects older people. In the case of implants, too, there is a lesser chance of these being rejected if they are encased in stem cells taken from the patient's own body. Kruse adds: "In the long term, we could possibly set up a cell bank for young people to store stem cells from their own sweat glands. They would then be available for use should the person need new cells – following an illness, perhaps, or in the event of an accident."