Researchers discover novel function of protein linked to Alzheimer's disease
A research team led by the National Neuroscience Institute (NNI) has uncovered a novel function of the Amyloid Precursor Protein (APP), one of the main pathogenic culprits of Alzheimer's disease. This discovery may help researchers understand how the protein goes awry in the brains of Alzheimer's disease patients, and potentially paves the way for the development of innovative therapeutics to improve the brain function of dementia patients.
The findings were published in the prestigious scientific research journal Nature Communications last month. The study, which is led by Dr Zeng Li and her team from NNI, involved investigators from Duke-NUS Graduate Medical School and the Agency for Science and Technology (A*STAR).
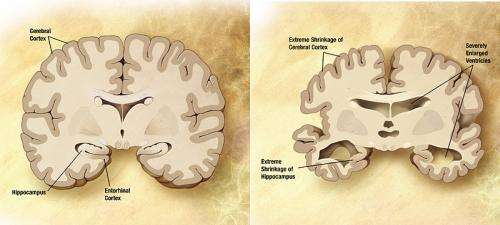
Alzheimer's disease is the most common form of dementia, which is set to rise significantly from the current 28,000 cases to 80,000 cases in 2030 among Singaporeans aged 60 and above. With a rapidly aging population, the burden of the disease will be profound affecting not just the person afflicted, but also the caregiver and family. While the exact cause of Alzheimer's disease remains unknown, one of its pathological hallmarks is clear – the clumping of APP product in the brain when the protein is abnormally processed.
Finding out more about APP can help researchers gain a better understanding of the disease, and potentially identify biomarkers and therapeutic targets for it. However up till this point, little was known about the APP's primary function in the brain.
The discovery – APP controls growth and maturation of brain cells
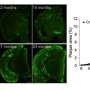
During this study, Dr Zeng and her team discovered that APP can control the growth and maturation of newborn brain cells, which are critical for the maintenance of a healthy brain function. APP does this by regulating a target known as microRNA-574-5-p. MicroRNAs are small molecules that influence the expression of human genes. The human body has many microRNAs to regulate the expression of different genes for proper cellular functions.
This study identified that microRNA-574-5p normally promotes the production of newborn neurons in the brain. In turn, the APP antagonises it to ensure the timely birth of new neurons to support normal brain function. In other words, the APP controls the growth and maturation of brain cells, without which neuron expression can go unregulated and cause brain activities to go haywire.
"Our findings highlight that microRNA-574-5p may be a potentially useful new target for drug development against Alzheimer's disease," said Dr Zeng Li, the principal investigator of the study. "We are just starting to understand how misregulated microRNA-574-5p expression can cause our brain activities to go wrong, and much more work needs to be done."
With this discovery, the research team intends to further their research by investigating the mechanisms of how the APP regulates microRNA-574-5p in association with the impairment of newborn neurons as seen in Alzheimer's disease. Eventually, they hope to develop the microRNA into a biomarker for the disease. And it does not just stop there – the finding also boosts the team's work on other neurodevelopmental conditions and brain disorders.
"Brain-specific microRNAs control neurogenesis during brain development, and their misregulation is implicated in other devastating psychiatric disorders like autism and schizophrenia," said Assistant Professor Shawn Je from Duke-NUS, a collaborator for this study. "So, this discovery also sheds light on ongoing collaboration work between our team and Dr Zeng's group to elucidate genetic and cellular mechanisms of autism."
Professor Stephen Cohen from A*STAR's Institute of Molecular and Cellular Biology, a world expert on microRNA biology, affirmed this discovery. He said, "This important study suggests a link between a key neurodegenerative disease gene and regulation of microRNAs in the brain. We are at an early stage of understanding how this microRNA might impact disease progression and associated behavior, but the prospects are exciting."