A simpler way to treat cancer

(Medical Xpress)—Sometimes the best solutions are the simplest ones. This is the philosophy that Northeastern professor Vladimir Torchilin and his team took in new research carried out in collaboration with physics professor Dmitri Lapotko from Rice University and presented in a paper published Sunday in the journal Nature Medicine.
"It's an absolutely new concept for treating cancer," said Torchilin, a Distinguished Professor of Pharmaceutical Sciences and director of the School of Pharmacy's Center for Pharmaceutical Biotechnology and Nanomedicine. "There are several factors combined in a very simple way, which results in a synergistic effect against the tumor."
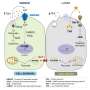
Together with the physicists at Rice, Torchilin's team created a cancer treatment approach that combines chemical and physical modalities to more efficiently destroy a tumor while leaving nearby healthy cells intact.
"You start with two very well known and simple things," Torchilin said. "Nanomedicines and nanoparticles." Both allow clinicians to steer materials to specific cells in the body using recognition factors that are disproportionately expressed on cancer cells. In the case of drug-loaded tumor-targeted nanomedicines, cancer cells welcome much higher concentrations of drug molecules. In the case of nanoparticles, it's tiny tumor-targeted crumbs of gold that end up inside the tumor cells.
Both methods are commonly used, but this is the first time they have been combined in a single therapeutic approach, explained Torchilin, who received the 2013 Blaise Pascal Medal for Biomedical Science from the European Academy of Science for his outstanding contribution to science and technology and the promotion of excellence in research and education.
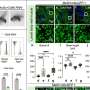
"So now you have normal cells with very few drug particles and gold particles, and cancer cells with a lot of those," he said. "When those particles come into the cell—just because of the way cells deal with foreign particles—they form clusters." In cancer cells, these clusters will be large while in normal cells they will be small.
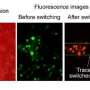
Next, the cells are irradiated with a laser beam, which interacts with such clusters and provokes a kind of a mini-explosion inside the cancer cell. Not only does this disrupt the physical structure of the cell from the inside out, it also breaks the nanomedicine delivery system containing the drug molecules, causing a simultaneous and massive drug release, Torchilin said.
"In normal cells with small clusters, the explosion won't happen or it will be very small so you cannot damage normal cells," he said. "But in a cancer cell you have a large cluster, so the bubble will be big, which can damage the cell. But, simultaneously, like any strong strike, it will release the drug."
Each part of the treatment is necessary and works in concert with the others to create concurrent chemical and physical attacks on the cancer cell, while leaving healthy cells relatively unharmed. "You use the standard things," Torchilin said. "You just use them all together. Simple."
More information: "On-demand intracellular amplification of chemoradiation with cancer-specific plasmonic nanobubbles." Ekaterina Y Lukianova-Hleb, et al. Nature Medicine (2014) DOI: 10.1038/nm.3484. Received 14 February 2013 Accepted 24 October 2013 Published online 01 June 2014