September 30, 2014 report
Axons growing out of dendrites? Neuroscientists hate when that happens
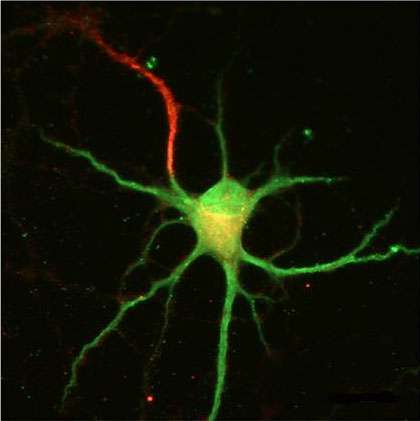
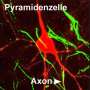
(Medical Xpress)—The well-behaved neuron receives signals through its many dendrites to generate spikes on a single axon. The electrical energy of these signals is generally believed to be integrated at the cell body and converted into pulses at the axon initial segment (this is the region where the axon sprouts, the AIS). There have always been exceptions to this neat and tidy picture of a neuron. Certain interneurons, dopaminergic cells, or neuroendicrine cells for example, have been shown to grow their axon out from a lucky dendrite instead of the soma. The existence of these anomalies is an inconvenient truth for all neuron modelers and typically they choose to ignore them. A paper recently published in Neuron now suggests that minimizing this fundamental neural character has been a mistake. Rather than mere quirk or curiousity the axon carrying dendrite, or AcD as the authors call it, is found on most neurons—at least among pyramidal cells in the CA1 area of the mouse hippocampus.
Many would argue about what percent of something the word "most" actually means. While it probably shouldn't be applied to situations where the majority is only 51 percent, if 20 out of the 36 cells you checked on had AcDs, it's probably at least a trend. The researchers had a look at the other parts of the hippocampus as well and found that about a quarter of the pyramidal cells there had AcDs too. It had been previously shown by other researchers that around 5% of CA1 pyramidal cells had axons shooting out of their apical dendrites. In other words, the upper regions of the extended cell. But using better imaging techniques the researchers now found that it is the basal dendrites below where the AcDs are more likely to be found.
Watching developing neurons under the time-lapse microscope as they pick one dendrite among many to crown as the axon is a source of wonderment for many neurobiologists. There is an inexhaustable supply of possible proteins to implicate in the process, and the game is never really over until it is over. If the whole dendritic tree, or at least the local parts are now fair game, the question of where exactly a neuron should put its axon just got real. Not only that, but where to keep the axon once established, or where to move it should the grass look greener, becomes even trickier.
Until now the axon was the one constant on which modelers could depend. There's no astrocytes in their models because astrocytes don't have axons. There are a few neural eunuchs floating around the invertebrate population (and in special places like the retina) with no axon to speak of, but for the most part they are a given in neurons. Unfortunately for the purely electrophysiological interpreation of cell structure, it now seems that a neuron placing its axon off in the electrically isolated world of a single basal dendrite would be the equivalent of growing your head out from your elbow.
The authors suggest there might be some method to this madness, namely that the particular dendrite that presumably controls the axon has certain useful "privileges". They used a precision technique called two-photon uncaging to activate just a few spines at a time in a select dendritic region of the cell. When they hit particular regions with a low intensity laser, glutamate could be released while they listend to the cell with a patch clamp electrode. They found that the AcDs could generate spikes at lower threshold and were also more synaptically excitable than dendrites without axons.
The researchers also looked at how far away from the soma the axon began, with particular emphasis on how long the initial segment region was. The protein markers used for this aren't always perfect, but in general there are a few good ones. Ankyrin-G highlights the axon while the AIS region can be identified with βIV-spectrin and the panNav antibody to Na channels. Spines are also identifyable and generally absent from the the AcD arm connecting to the soma. The length of the AIS portion is important for several reasons. In the avian auditory system for example, the length and location of the AIS varies widely and tunes the cell to certain frequencies.
The myelin-free AIS was the subject of a fairly impactful study we recently covered here. A graded distribution of the AIS length was found in the cortex with the more superficial cells having longer "open" axon. The morphology of the AIS region pinpointed in all of these types of studies undoubtedly plays a critical role in the kinds of spikes a cell can generate. While inconsequential to most models, things like the refractory period, frequency, size and shape of the spike may all be controlled in part by the AIS.
In rodent CA1 neurons, the dendrites undergo significant differention and maturation during the postnatal to sexual maturity period. The researchers found that most of the AcDs were formed during this same period. While these studies are painstaking and well executed, I'm not sure that these kinds of things are really telling us all that much. The time from birth to maturation is hardly a "when" and definitely not much of a "why". The question I would have is if neurons are largely ignoring most of the "electrotonic" real estate of their larger dendtiric tree in generating spikes, than perhaps integrating small potentials is not the main concern of dendrites.
In the last few months we have been made aware that neural pulses can go higher, faster, further in ways we may not have once figured. Spikes can pass through each other unscathed, and they can also be generated at fantastic rates above 1500 hz, in addition to being sent in either direction. Undoubtedly some of these new features extend to dendrites. But considering the high cost usually associated with synaptic activity it seems that holding on to several hardly-used dendritic showrooms would be a bit inefficient if the axon never in fact moves around to inhabit them.
Perhaps it does. Perhaps one day we might even catch a neuron in transient possesion of two axons, transferring an axon to a neighboring cell, or transitioning from bipolar to a pseudo-unipolar morphology. In either case some observers may asign the change to events in the nucleus, or maybe to the activites of neighboring cells. It may not however, be such an imaginative stretch to asign responsibility for morphological transitions to the activites of the mitochondria. For example, a neuron found to have high mitochondrial heteroplasmy (mitochondria with different genetic makeup obtained either from other cells or from its own original lineage traced to the germline) might have ways to segregate them throughout the cell—one way being to isolate their transport or flow to the axon.
The observation that axons can and do transexudate questionable mitochondria far away from the soma could even shed light on the primal urge for the neuron to extend an axon across incredible voids to parts unknown in the first place. Clearly speculation, but perhaps of the kind needed to make progress here.
More information: Axon-Carrying Dendrites Convey Privileged Synaptic Input in Hippocampal Neurons, Neuron, Volume 83, Issue 6, p1418–1430, 17 September 2014. dx.doi.org/10.1016/j.neuron.2014.08.013
© 2014 Medical Xpress