First human in vitro model of rare neurodegenerative condition created
Researchers at University of California, San Diego School of Medicine and Rady Children's Hospital-San Diego have created the first stem cell-derived in vitro cellular model of a rare, but devastating, neurodegenerative condition called Cockayne syndrome (CS).
The findings are published in the current online issue of the journal Human Molecular Genetics.
CS is characterized by short stature and an appearance of premature aging. Traits include extreme sensitivity to sunlight, abnormally small head size (microcephaly), impaired development of the nervous system, eye problems and a failure to gain weight and grow at the expected rate, called "failure to thrive." While specific symptoms of CS may be treatable, there is no cure and prognosis is poor. Most patients do not survive past their twenties.
Eighty percent of CS cases are caused by mutations in a gene called ERCC6, with codes for a protein called Cockayne Syndrome B (CSB). Past efforts to model the neurological aspects of the disease have proved difficult because existing mouse models do not display classical neurological symptoms.
"What we have needed is a robust human in vitro cellular model - a so-called 'disease in a dish' - that would allow us to understand fundamentally what is happening and which could point us toward possible therapeutic targets and treatments," said senior author Alysson R. Muotri, PhD, associate professor in the UC San Diego School of Medicine departments of Pediatrics and Cellular and Molecular Medicine.
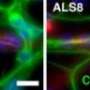
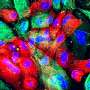
Muotri and colleagues have previously used induced pluripotent stem cells to create neuronal models of autism spectrum disorders. Mirroring those earlier efforts, Muotri and his team generated the in vitro CS cell model by taking skin cells from two individuals with CS, reverting them to induced pluripotent stem cells, then reprogramming the stem cells to differentiate as neural progenitor cells and neurons. The neurons subsequently formed functioning networks, popularly dubbed "mini-brains."
The CS networks allowed researchers to identify areas of cellular dysfunction compared to normal neuronal networks from control models. Specifically, they noted that CSB-deficient neuronal networks displayed altered electrophysiological activity, including diminished ability to grow synaptic connections to other neurons and synchronize activities.
"This work provides an alternative to existing models for studying neural-specific aspects of CS," said Muotri. "It reveals that CSB protein is required for normal neuronal function. It shows the feasibility of constructing a new and relevant human in vitro model with potential clinical interest."
More information: Human Molecular Genetics, dx.doi.org/10.1093/hmg/ddw008