February 23, 2016 report
Probiotics dramatically modulate liver cancer growth in mice

(Medical Xpress)—Medical research over the last decade has revealed the effects of the gut microbiome across a range of health markers including inflammation, immune response, metabolic function and weight. There is a corresponding interest in priobiotics as a therapeutic approach to the many illnesses affected by gut flora.
Previous studies have demonstrated the beneficial role of probiotics in reducing gastrointestinal inflammation and preventing colorectal cancer, but a new study published in the Proceedings of the National Academy of Sciences explored their immunomodulatory effects on extraintestinal tumors: specifically, in hepatocellular carcinoma (HCC).
HCC is the most common type of liver cancer, and though it is relatively uncommon in the United States, it's the second-most deadly type of cancer worldwide and is particularly prevalent in regions with high rates of hepatitis. Because low-grade tumors are difficult to detect, and high-grade tumors have poor prognosis, medical researchers are interested in alternative and complementary therapeutic approaches to improve mortality rates.
The researchers designed a study in a mouse model of HCC that quantified the immunological effects of a novel probiotic formulation called Prohep. They fed the mice Prohep for a week prior to tumor inoculation, and they observed a 40 percent reduction of tumor weight and size compared with control animals. Further, they established that the beneficial effects of the probiotics were closely related to the abundance of beneficial bacteria promoted by Prohep. These bacteria produce anti-inflammatory metabolites, which regulated pro-inflammatory immune cell populations via crosstalk between the gut and the liver tumor.
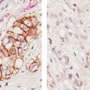
Among their findings, the researchers report that the probiotics reduced liver tumor growth by inhibiting angiogenesis, the process by which the body generates new blood vessels from existing ones, which is essential for tumor growth. They found significantly raised levels of hypoxic GLUT-1+, indicating that tumor reductions were due to hypoxia caused by reduced blood flow. Further, the tumors in the treated mice had 52 percent lower blood vessel area and 54 percent fewer vessel sprouts than the untreated mice.
They also determined that Prohep treatment down-regulated IL-17, a pro-inflammatory angiogenic factor. Because HCC is a highly vascularized tumor, the cancer is generally associated with high levels of IL-17 and an immune T-cell called T helper 17 (Th17), which is transported from the gut to HCC tumors via circulation. The researchers believe that reduced Th17 in tumor cells impedes the inflammation and angiogenesis and limits tumor growth.
It's not surprising that they also found that probiotics increased the anti-inflammatory bacteria and metabolites present in the guts of treated mice. They conclude that Prohep intake has the capability of inhibiting tumor progression by modulating the gut microbiota.
They write, "In conclusion… Prohep was effective in reducing HCC growth significantly in mice. Th17 was likely to be the major producer of IL-17 in the tumor microenvironment that has linked to HCC growth and angiogenesis, and its decrease in tumor was probably related to the reduced recruitment from the gut via circulation. The antitumour function offered by Prohep was likely to associate with modulation of the gut microbiota by inducing the secretion of anti-inflammatory IL-10 cytokine and suppressing Th17 cell differentiation in the gut."
More information: Probiotics modulated gut microbiota suppresses hepatocellular carcinoma growth in mice. PNAS 2016 ; published ahead of print February 16, 2016, DOI: 10.1073/pnas.1518189113
Abstract
The beneficial roles of probiotics in lowering the gastrointestinal inflammation and preventing colorectal cancer have been frequently demonstrated, but their immunomodulatory effects and mechanism in suppressing the growth of extraintestinal tumors remain unexplored. Here, we adopted a mouse model and metagenome sequencing to investigate the efficacy of probiotic feeding in controlling s.c. hepatocellular carcinoma (HCC) and the underlying mechanism suppressing the tumor progression. Our result demonstrated that Prohep, a novel probiotic mixture, slows down the tumor growth significantly and reduces the tumor size and weight by 40% compared with the control. From a mechanistic point of view the down-regulated IL-17 cytokine and its major producer Th17 cells, whose levels decreased drastically, played critical roles in tumor reduction upon probiotics feeding. Cell staining illustrated that the reduced Th17 cells in the tumor of the probiotic-treated group is mainly caused by the reduced frequency of migratory Th17 cells from the intestine and peripheral blood. In addition, shotgun-metagenome sequencing revealed the crosstalk between gut microbial metabolites and the HCC development. Probiotics shifted the gut microbial community toward certain beneficial bacteria, including Prevotella and Oscillibacter, that are known producers of antiinflammatory metabolites, which subsequently reduced the Th17 polarization and promoted the differentiation of antiinflammatory Treg/Tr1 cells in the gut. Overall, our study offers novel insights into the mechanism by which probiotic treatment modulates the microbiota and influences the regulation of the T-cell differentiation in the gut, which in turn alters the level of the proinflammatory cytokines in the extraintestinal tumor microenvironment.
© 2016 Medical Xpress