Hidden genetic mutations in stem cells could undermine therapeutic benefit
For the first time, scientists have confirmed the long-standing hypothesis that as people age, they accumulate gene mutations in their mitochondria—cells' energy source.
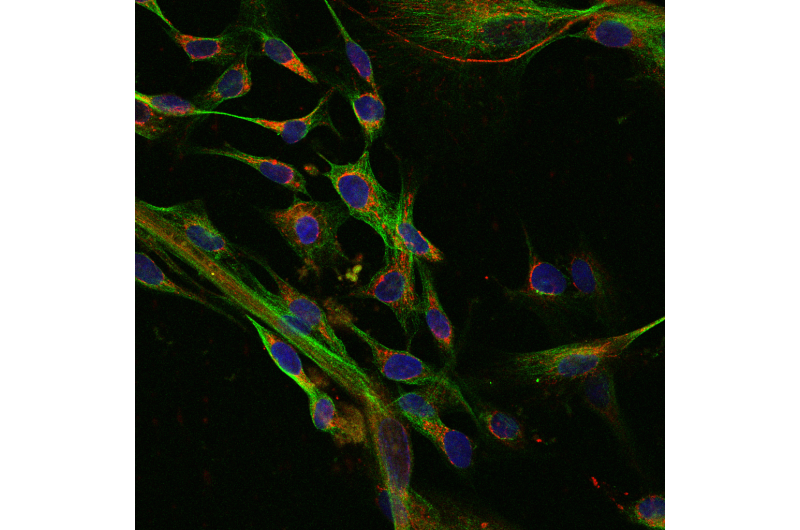
A team led by Shoukhrat Mitalipov, Ph.D., director of the Center for Embryonic Cell and Gene Therapy at Oregon Health & Science University, has discovered that induced pluripotent stem, or iPS, cells, a type of stem cell derived from patients' skin or blood cells contain faulty mitochondrial DNA. The study was published today in the journal Cell Stem Cell.
"Pathogenic mutations in our mitochondrial DNA have long been thought to be a driving force in aging and age-onset diseases, though clear evidence was missing. Now with that evidence at hand, we know that we must screen stem cells for mutations or collect them at younger age to ensure their mitochondrial genes are healthy," said Mitalipov. "This foundational knowledge of how cells are damaged in the natural process of aging may help to illuminate the role of mutated mitochondria in degenerative disease."
Mitochondrial genes reside outside of the nucleus and have been known to be prone to damage. Mutations in mitochondrial DNA, which arise randomly within individual cells as we age, can limit cells' ability to create energy, produce signals and function properly.
Potential therapies using stem cells hold tremendous promise for treating human disease. However, defects in the mitochondria could undermine the iPS cells' ability to repair damaged tissue or organs. To avoid impairing IPS cells' therapeutic value, Mitalipov and colleagues recommend screening the cells for mitochondrial DNA mutations.
"If you want to use iPS cells in a human, you must check for mutations in the mitochondrial genome," said Taosheng Huang, M.D., Ph.D., a medical geneticist and director of the Mitochondrial Disorders Program at Cincinnati Children's Hospital Medical Center. "Every single cell can be different. Two cells next to each other could have different mutations or different percentages of mutations."
"This collaborative multi-disciplinary effort identifies 'mitochondrial genome integrity' as a vital readout in assessing the proficiency of patient-derived regenerative products destined for clinical applications", adds study co-author Andre Terzic, M.D., Ph.D., Director, Mayo Clinic Center for Regenerative Medicine.
Every individual has trillions of cells and thousands of copies of mitochondria in each cell. As cells age, thousands of different mutations in each mitochondrial genome are possible. Because each cell has its own unique mutation, when researchers examine a pinch of skin or drop of blood containing millions of cells, most mitochondrial mutations are hidden. The mutation would only be visible if every cell has the same mutation.
However, these mutations become detectable in iPS cells. In the process of making iPS cell lines, researchers expand clones from each individual patient skin or blood cell. As a result, every cell in the iPS cell line will contain the same mitochondrial DNA mutations as that initial adult cell.
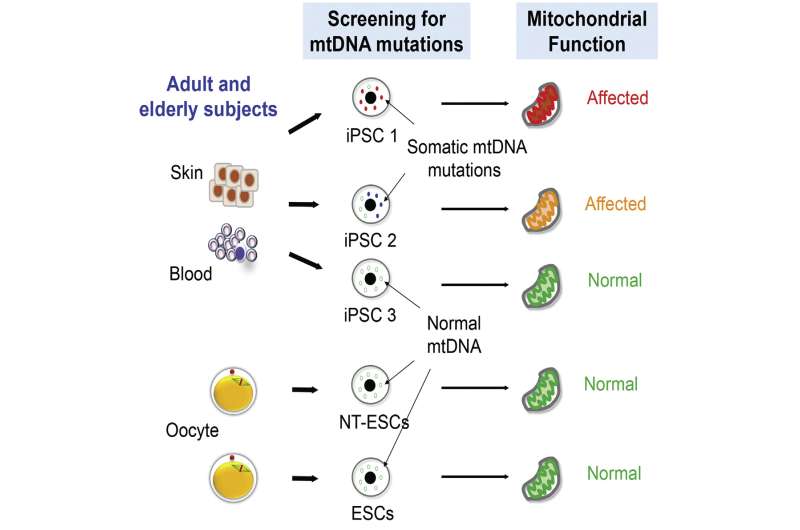
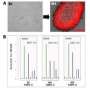
In this study, rather than studying one iPS cell line, the researchers derived and sequenced 10 iPS cell clones from each patient tissue sample to get a better understanding of mitochondrial DNA mutation rates.
They took samples of blood and skin from healthy people and people with degenerative diseases, ranging in age from 24 to 72. When they tested the pooled skin or blood cells for mitochondrial DNA mutations, the levels of mutations appeared low.
Then they profiled 20 iPS cell lines per patient - 10 from their blood cells and 10 from their skin cells. When they sequenced the iPS cell lines, they found higher numbers of mitochondrial DNA mutations, particularly in cells from patients older than 60. They analyzed 130 iPS cell lines and discovered 80 percent showed mutations. They also found higher percentages of mitochondria containing mutations within a cell. The higher the load of mutated mitochondrial DNA in a cell, the greater the cell's function is compromised.
The authors recommend producing and screening multiple lines per patient and then choose the least damaged line.
In May 2013, Mitalipov was the first scientist in the world to demonstrate the successful use of somatic cell nuclear transfer, or SCNT, to produce human embryonic stem cells from an individual's skin cell. That breakthrough was one of a six-year chain of discoveries that included his 2007 work demonstrating the nuclear transfer method to create embryonic stem cells from a nonhuman primate. OHSU scientists have also demonstrated that SCNT allows replacement of mutated mitochondrial genes with healthy donor egg mitochondria while retaining the patient cell's nucleus.
More information: "Age-Related Accumulation of Mitochondrial DNA mutations in Adult-Derived Human iPSCs," Cell Stem Cell, 2016. dx.doi.org/10.1016/j.stem.2016.02.005