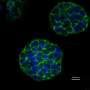
Three-dimensional model sheds light into the synergy between cell microenvironment and cancer stem cells

The fight against deadly forms of brain cancer, such as glioblastoma, could soon benefit from a major breakthrough, thanks to A*STAR researchers. The team has found a new link between cancer cell growth and its microenvironment.
Stem cells in tumors drive the development and drug resistance of certain cancers, including brain cancers, which thwarts anti-cancer treatments. Their stemness, or ability to grow, self-renew, and differentiate into various cancer cells, is influenced by their microenvironment. This accounts for interactions with other cells, adhesion molecules, and the matrix surrounding the cells, also called extracellular matrix.
In addition to acting as a support for cells, the extracellular matrix mediates cell signaling and is integral in promoting cancer cell stemness. However, studies have focused on signaling pathways within cells to assess cancer stemness, so the role of the microenvironment is poorly understood.
To clarify the contribution of the extracellular matrix to cancer stemness, a team led by Andrew Wan from the A*STAR Institute of Bioengineering and Nanotechnology used an approach involving a three-dimensional (3D) model for brain cancer. In this model, entangled electrospun polystyrene fibers provided a porous scaffold for cell culture. The fibers were coated with specific extracellular matrix proteins known as laminins.
The researchers cultivated cells from an aggressive type of brain tumor, glioblastoma, in the 3D model and two-dimensional (2D) conventional polystyrene tissue culture plates and monitored their growth (see image). Gene and protein expression studies revealed that the 3D culture system boosted cancer stemness to a greater extent than its 2D equivalent.
Wan's team further discovered that the 3D system and extracellular matrix worked together to stimulate stemness. Specifically, they found evidence that two integrins—proteins that reside in glioblastoma cell membranes—promoted the formation of cancer stem cell-like cells by interacting with the laminins.
Co-workers from the National Neuroscience Institute corroborated these observations by analyzing patient glioma databases through computational approaches. "Our collaborators showed that these same proteins were often expressed at higher levels in more aggressive brain tumors," says Wan.
These findings provide a new angle for future approaches to anti-cancer therapies, explains Wan. "We were excited about the results as we had not expected such a significant effect when we started our experiments," he notes.
Wan's team is currently investigating the process through which interactions between brain cancer cells and extracellular matrix give rise to stemness in 3D culture systems. "An understanding of how that happens may one day aid in tackling the problem of glioblastoma," he adds.
More information: Nina K.L. Ma et al. Collaboration of 3D context and extracellular matrix in the development of glioma stemness in a 3D model, Biomaterials (2016). DOI: 10.1016/j.biomaterials.2015.11.031