Research identifies a molecular basis for common congenital brain defect
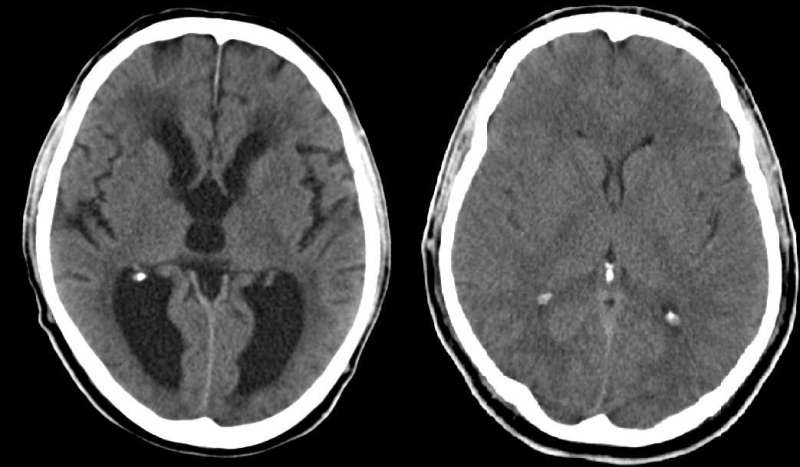
Scientists at Sanford Burnham Prebys Medical Discovery Institute (SBP) have discovered a molecular cause of hydrocephalus, a common, potentially life-threatening birth defect in which the head is enlarged due to excess fluid surrounding the brain. Because the same molecule is also implicated in Down's syndrome, the finding, published today in the Journal of Neuroscience, may explain the ten-fold increased risk of hydrocephalus in infants born with Down's.
"We found that deleting the gene for sorting nexin 27, or SNX27, which plays a major role in the development of Down's syndrome, causes hydrocephalus," said Huaxi Xu, Ph.D., the Jeanne and Gary Herberger Leadership Chair of SBP's Neuroscience and Aging Research Center. "The mechanism we uncovered likely only accounts for a fraction of hydrocephalus cases, but we identified potential non-surgical treatments for these cases that deserve further study."
Hydrocephalus affects one or two of every 1,000 births. Some causes of hydrocephalus are known, including several well-characterized brain and skull malformations that block fluid outflow, but it can also arise in the absence of other obvious abnormalities. The condition is treated by surgically inserting a shunt to divert the fluid to another part of the body where it can be absorbed. However, these tubes can become infected, and about half the time, they fail, causing headaches, vomiting, fever, and irritability until the shunt is replaced.
The new study followed up on prior results from Xu's lab showing that SNX27, a protein that regulates traffic of other proteins within cells, is found at lower than normal levels in the brains of individuals with Down's syndrome. They also found that inactivating the gene for SNX27 in mice causes learning and memory problems similar to those in Down's.
Here, Xu's team looked at overall brain development in mice without SNX27. They observed severe hydrocephalus, with fluid-filled cavities (ventricles) in the brain that were much larger than normal. Examining potential causes, they saw that these mice lacked the cells that normally line the ventricles and circulate fluid in the brain, called ependymal cells.
The researchers also determined why ependymal cells aren't generated—without SNX27, brain stem cells generate too much of the active form of a protein called Notch that keeps them from becoming ependymal cells. The active form of Notch is created by an enzyme called gamma-secretase, whose activity is regulated by SNX27. Without SNX27, too much gamma-secretase remains active.
"Proper flow of fluid out of the brain isn't just crucial in brain development—it also helps eliminate toxic proteins such as amyloid beta, which causes Alzheimer's," added Xu. "Since we've already shown that lack of SNX27 increases production of amyloid beta, genetic variants that cause lower than normal levels of SNX27 would greatly increase risk for Alzheimer's. This double effect likely explains why Down's syndrome patients' brains exhibit Alzheimer's pathology by adulthood."
Wang, Xu, and their collaborators went on to show that giving either a drug that inhibits gamma-secretase to SNX27-deficient mice prevents them from developing hydrocephalus.
"Gamma-secretase inhibitors could be a future treatment for cases of hydrocephalus caused by ependymal cell defects," commented Xu. "However, further study is required to determine whether this approach is relevant to humans."
More information: Journal of Neuroscience, DOI: 10.1523/JNEUROSCI.1620-16.2016