Potential new cancer treatment activates cancer-engulfing cells
Macrophages are a type of white blood cell that can engulf and destroy cancer cells. A research group led by Professor MATOZAKI Takashi, Associate Professor MURATA Yoji, and YANAGITA Tadahiko (Kobe University Department of Biochemistry and Molecular Biology, Division of Molecular and Cellular Signaling), discovered that by using an antibody for a particular protein found on macrophages, the macrophage is activated, and cancer cells are effectively eliminated. This discovery could lead to the development of new cancer treatments. The findings were published on January 12 in the online version of JCI Insight.
Cancer is a leading cause of death worldwide. In recent years, attention has been drawn by the effectiveness of treatment that targets specific proteins that express on both cancer cells and healthy cells including immune system cells (targeted therapy). However, issues with this treatment include the emergence of cancer cells resistant to targeted drugs, and side effects which differ from those for conventional anticancer drugs.
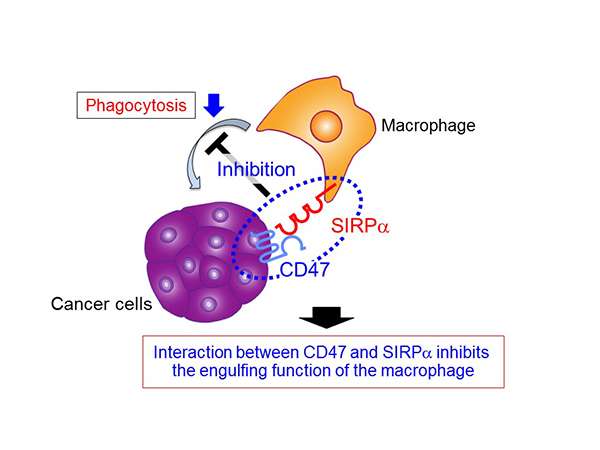
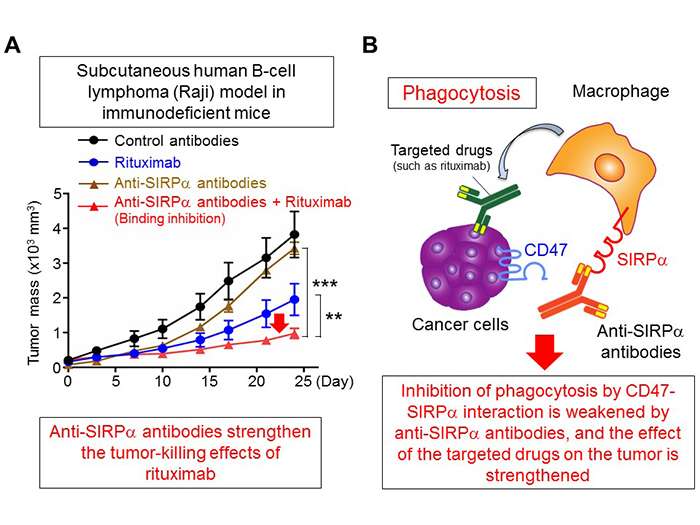
In their previous research, Professor Matozaki's research group discovered that when the protein SIRPα, which is expressed on the cell membrane of macrophages, interacts with the protein CD47 which is expressed on the cell membrane of engulfment targets (such as cancer cells and senescent cells), the engulfment ability (phagocytosis) of the macrophages is inhibited (figure 1). This time, using immunodeficient mice subcutaneously injected with Raji cells (derived from human B-cell lymphoma), the group discovered that an anti-SIRPα antibody, which specifically reacts with SIRPα and inhibits the interaction of CD47 and SIRPα, helps a target drug known as rituximab to suppress tumor growth (figure 2A). Furthermore, they demonstrated that based on anti-SIRPα antibodies that inhibited CD47-SIRPα interaction, the phagocytic properties of macrophages were strengthened by rituximab. The researchers concluded that anti-SIRPα antibodies, which inhibit CD47-SIRPα interaction, could be highly effective against tumors by strengthening the phagocytosis of macrophages for cancer cells using target drugs such as rituximab (figure 2B).
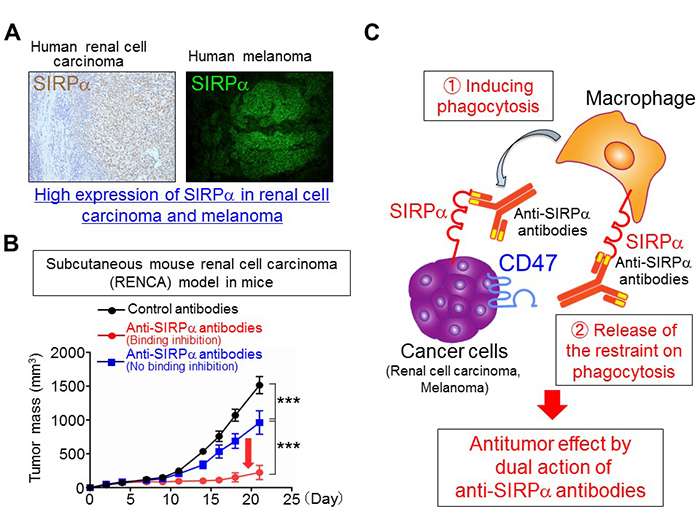
The research group also discovered that both renal cell carcinoma and melanoma express high levels of SIRPα in humans and mice (figure 3A). In mice injected with mouse renal cell carcinoma (RENCA) cells or melanoma cells, they found that the sole administration of anti-SIRPα antibodies suppressed the growth (figure 3B) or metastasis of tumors. However, the suppression of tumor growth by anti-SIRPα antibody treatment was weakened in mice whose macrophages had been eliminated. In addition, from phagocytosis experiments using macrophages and renal cell carcinoma cells or melanoma cells, they showed that anti-SIRPα antibodies (1) induce phagocytosis of cancer cells by macrophages through the direct binding to SIRPα on cancer cells; (2) prevent the CD47-SIRPα interaction between cancer cells and macrophages, removing the restraints on phagocytosis of macrophages (activating the macrophages). Therefore, they concluded that the antitumor effects of anti-SIRPα antibodies on SIRPα-expressing cancer cells are based on this dual action (figure 3C).
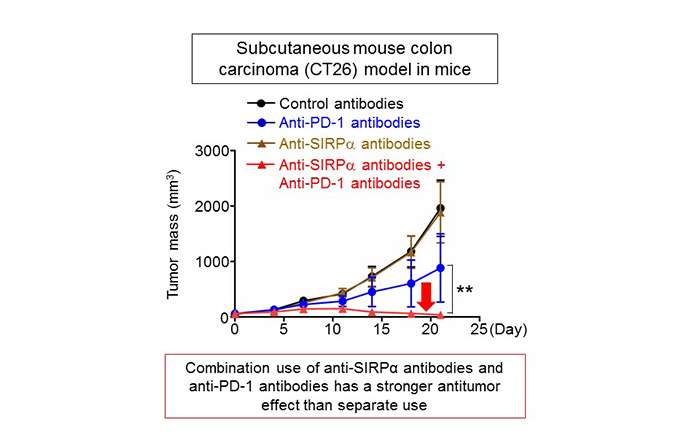
Recently, anti-PD-1 antibodies that inhibit the functions of the PD-1 protein have demonstrated efficacy as anticancer drugs. The PD-1 protein is one of the immune checkpoint molecules(6) for cytotoxic T-cells. In mice subcutaneously implanted with mouse colon cancer cells (CT26 cells), the simultaneous administration of anti-SIRPα antibodies and anti-PD-1 antibodies has a stronger effect on inhibiting tumor growth compared to their separate administration (figure 4). The details of the action mechanism for anti-SIRPα antibodies are not fully understood, but simultaneous use of immune checkpoint inhibitors such as anti-PD-1 antibodies that act against cytotoxic T-cells and anti-SIRPα antibodies has a stronger antitumor effect than their separate use.
These findings have clarified that the antibodies for SIRPα, an immune checkpoint molecule for macrophages related to the innate immune system function, can be used as a new cancer treatment. In the future, as well as analyzing the mechanism for the antitumor effect of anti-SIRPα antibodies in more detail, while carefully assessing the safety of the treatment, anti-SIRPα antibodies can potentially be developed as anticancer drugs.
More information: Tadahiko Yanagita et al, Anti-SIRPα antibodies as a potential new tool for cancer immunotherapy, JCI Insight (2017). DOI: 10.1172/jci.insight.89140