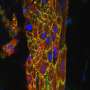
Heart tissues of different origins can 'beat' in sync

Researchers from MIPT and the University of Bonn have shown that heart tissues of different origins can contract in sync. In a series of experiments, they first merged two rat tissues of different ages and then combined rat and mouse tissue. Excitation waves were transmitted successfully from one tissue to another, which theoretically means that artificially grown heart patches can fit in with excitable cardiac tissues. The paper was published in Biomaterials Science.
Professor Konstantin Agladze, who heads the Laboratory of the Biophysics of Excitable Systems at MIPT, says, "People are only now discussing growing cardiac patches. And the question is whether we pursue these engineered implantable tissue constructions, considering that they could fail to merge into one excitable tissue with the heart. We resolve this issue by showing that merging into one excitable tissue is, indeed, possible, even if we're dealing with cells from different animals, let alone cultures of merely different ages. These two species, rats and mice, are related—but so are humans and apes."
Repairing the heart
Researchers working in the field of cardiac regenerative medicine are hopeful about the possible application of cultured patches. In theory, a piece of cultured heart tissue could be used to repair regions of the heart damaged as a result of an infarction. However, this has never been accomplished experimentally. Transplanted cells died within several days, with little to no improvement observed. An important property of cardiac tissue, which actually allows the heart to beat, is its ability to contract in response to an electrical signal. Until now, it has remained unknown whether a piece of cultured tissue can merge with host cardiac tissue and function correctly. For the heart muscle to contract, an electrical signal must be transmitted between the cells. But there was no solid proof that implantation of foreign cells into the heart tissue would result in an electrical coupling with host cells, which is needed to create an integrated system. If there is no coupling, then cultured patches are useless in terms of repairing damaged heart tissue. The researchers set out to test if it is even possible for an electrical excitation to be transmitted between two tissues of different origins. To do this, they grew cardiac tissue in a container designed specifically for this experiment.
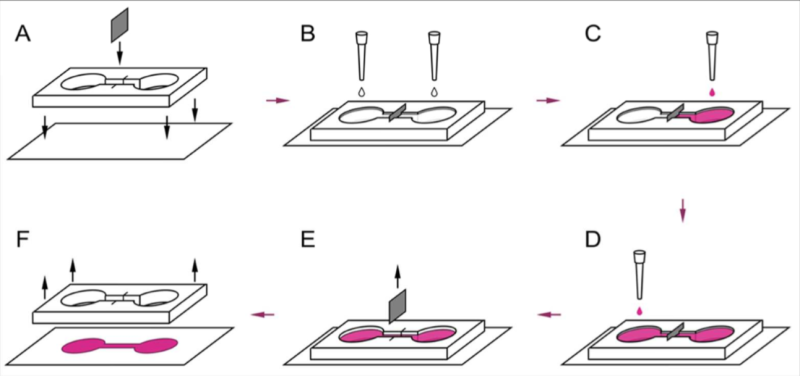
Experimental design
The researchers designed a dumbbell-shaped container made up of two circular compartments—five millimeters in diameter each—connected by a thin, seven-millimeter-long channel. To prevent the cells of different cultures from mixing with each other, a partition was inserted halfway through the channel. Cells of the first culture were seeded to one of the compartments. Then, after about an hour, cells of the second culture were seeded to the other compartment. When the container was filled with cells on both sides, the partition was removed. This allowed the two tissues to merge at the point where the partition had been. The researchers stimulated one tissue to see if the excitation wave would be transmitted to the other tissue. To observe wave propagation, they introduced a fluorescent dye called Fluor-4 into the container. Excitation waves cause this chemical to emit light that can be recorded using a high-sensitivity camera.
Different tissues
The first experiment featured heart cells of neonatal rats. The primary cell culture was first seeded into one compartment and then, three days later, into the other. This means that the two tissues were at different stages in their development. Still, when one of them was stimulated with an electrode, the excitation wave was transmitted across and beyond the border between the two tissues. The researchers then decided to see if the experimental results could be reproduced using cells from two different species, namely rats and mice. Although the excitation wave did propagate through both tissues, its speed differed between the two cultures.
A further test to be on the safe side
It is theoretically possible that cells not directly stimulated with an electrode could be excited by the electric field of the electrode. To rule out this scenario, the researchers performed a further series of experiments. They used genetic engineering methods to insert a light-sensitive protein called channelrhodopsin-2 into mouse cells, which were then exposed to light. Despite rat cells not being sensitive to light, they, too, conducted the excitation wave when mouse cells were illuminated. This proves conclusively that the two different tissues are, in fact, electrically coupled to each other. In addition, the synchronization of different cultures was confirmed at the micro-level when individual cells were studied.
It should be noted, however, that certain peculiarities were observed at the border between the two cultures, including partial blocking of excitation waves crossing the border at certain frequencies. The next question that needs to be addressed before growing cardiac patches is whether these anomalies could increase the risk of arrhythmia.
More information: N. N. Agladze et al, Synchronization of excitable cardiac cultures of different origin, Biomater. Sci. (2017). DOI: 10.1039/C7BM00171A