New findings point to potential therapy for Parkinson's Disease
A new study, published in Proceedings of the National Academy of Sciences (PNAS), sheds light on a mechanism underlying Parkinson's disease and suggests that Tacrolimus—an existing drug that targets the toxic protein interaction explored in the study—could be used as a novel treatment.
Gabriela Caraveo Piso, Ph.D., assistant professor of Neurology in the Division of Movement Disorders, was the first and corresponding author of the study.
Parkinson's disease is a progressive neurological disorder marked by the loss of dopaminergic neurons. There is no cure, and current treatments to help control the symptoms of Parkinson's—including tremor, slow movement and loss of balance—are limited.
Although the fundamental cause of Parkinson's disease is unknown, dysfunction of a protein called alpha-synuclein has long been thought to play an important role.
Previous investigations by Caraveo Piso and collaborators had established that increased activity of an enzyme called calcineurin is associated with the alpha-synuclein toxicity seen in Parkinson's.
In the current study, the scientists built upon those discoveries to demonstrate, for the first time, that a protein called FKBP12 is a major regulator of calcineurin activity, and thus contributes to alpha-synuclein toxicity.
"Our new mechanistic findings have revealed that FKBP12 is the protein that tunes calcineurin activity," Caraveo Piso said. "It's very reminiscent of the Goldilocks story: In the context of alpha-synuclein, both too much and no calcineurin activity are detrimental—but an intermediate level of activity is protective."
The scientists also investigated whether reducing the functional interaction between calcineurin and FKBP12 might be protective against alpha-synuclein toxicity in vivo.
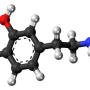
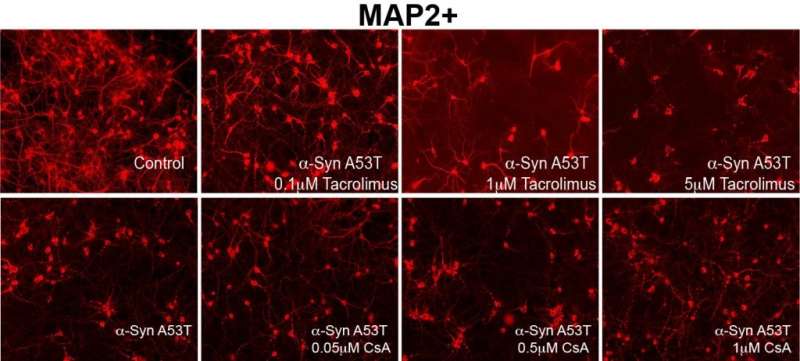
To do so, they employed an already well-established, FDA-approved inhibitor of this interaction, called Tacrolimus. The drug is currently used clinically in high doses to prevent organ rejection after a transplant, a process in which calcineurin also plays a critical role.
In an animal model of Parkinson's disease, the investigators demonstrated that Tacrolimus—when administered in low doses to achieve partial reduction of calcineurin activity—led to improvements in the functional features of dopaminergic neurons, and hence behavioral phenotypes.
Caraveo Piso noted that the discovery has strong potential for future clinical applications.
"We provide robust animal evidence for a feasible treatment against Parkinson's by repurposing Tacrolimus at low, sub-immunosuppressive doses," Caraveo Piso said. "Since Tacrolimus has high brain penetrance and can be given safely, a clinical trial of Tacrolimus in patients with early-stage Parkinson's would likely pose no serious logistical or regulatory difficulties."
The findings might also have implications for other related neurological diseases, such as Alzheimer's, Caraveo Piso notes, an area where calcineurin also plays an important role.
More information: Gabriela Caraveo et al. FKBP12 contributes to α-synuclein toxicity by regulating the calcineurin-dependent phosphoproteome, Proceedings of the National Academy of Sciences (2017). DOI: 10.1073/pnas.1711926115