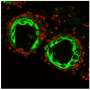
Hairy skin grown from mouse stem cells
Indiana University School of Medicine researchers have cultured the first lab-grown skin tissue complete with hair follicles. This skin model, developed using stem cells from mice, more closely resembles natural hair than existing models and may prove useful for testing drugs, understanding hair growth, and reducing the practice of animal testing. The work appears January 2 in the journal Cell Reports.
Although various methods of generating skin tissue in the lab have already been developed, their ability to imitate real skin falls short. While real skin consists of 20 or more cell types, these models only contain about five or six. Most notably, none of these existing skin tissues is capable of hair growth.
Karl Koehler, an assistant professor of otolaryngology at the Indiana University School of Medicine, originally began using pluripotent stem cells from mice, which can develop into any type of cells in the body, to create organoids—miniature organs in vitro—that model the inner ear. But Koehler and his team discovered they were generating skin cells in addition to inner ear tissue, and their research shifted towards coaxing the cells into sprouting hair follicles.
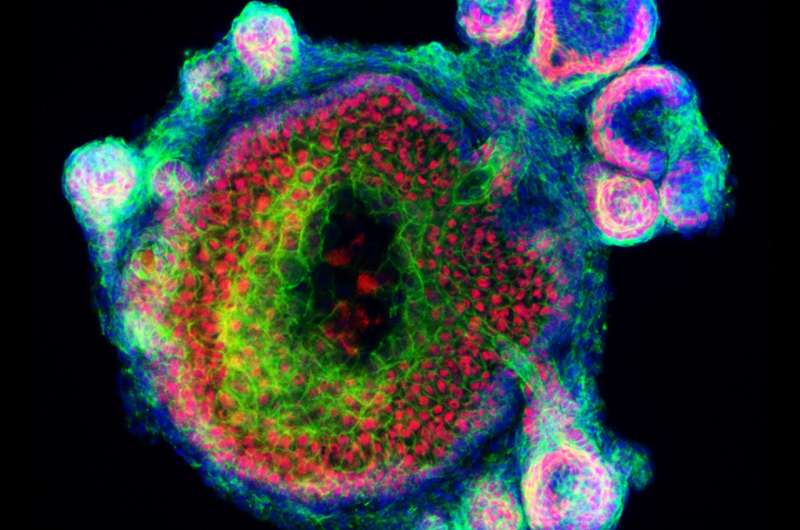
The team's recent research demonstrates that a single skin organoid unit developed in culture can give rise to both the epidermis (upper) and dermis (lower) layers of skin, which grow together in a process that allows hair follicles to form the same way as they would in a mouse's body.
"You can see the organoids with your naked eye," Koehler says. "It looks like a little ball of pocket lint that floats around in the culture medium. The skin develops as a spherical cyst, and then the hair follicles grow outward in all directions, like dandelion seeds."
While the researchers were unable to identify exactly which types of hairs developed on the surface of the organoid, they believe the skin grew a variety of hair follicle types similar to those present naturally on the coat of a mouse. The skin organoid itself consisted of three or four different types of dermal cells and four types of epidermal cells—a diverse combination that more closely mimics mouse skin than previously developed skin tissues.
By observing the development of this more lifelike skin organoid, the researchers learned that the two layers of skin cells must grow together in a specific way in order for hair follicles to develop. As the epidermis grew in the culture medium, it began to take the rounded shape of a cyst. The dermal cells then wrapped themselves around these cysts. When this process was disrupted, hair follicles never appeared.
"One thing we explored in the paper is that if we destroy the organoids and try to put them back together, they don't always generate hair follicles," Koehler says. "So, we think that it's very important that the cells develop together at an early stage to properly form skin and hair follicles."
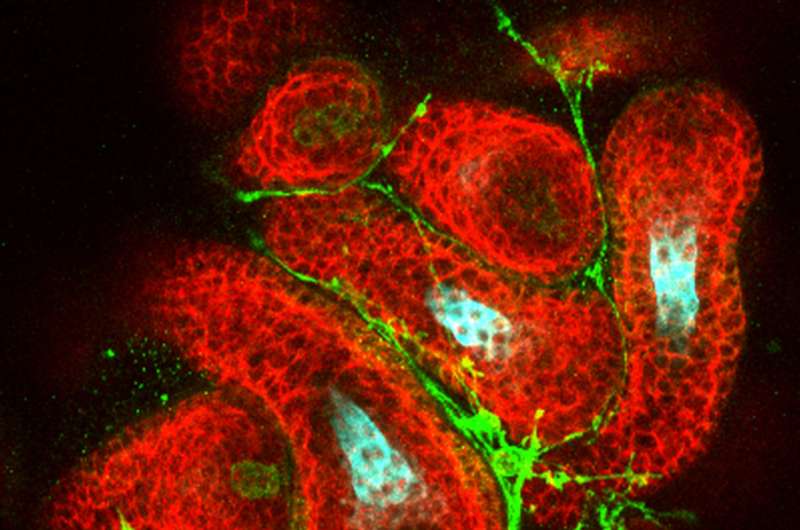
After discovering this recipe for lab-grown hair follicles, the researchers must now work to overcome a new roadblock in the study of in vitro hair development—physical limitations that prevent the hairs from shedding and regenerating. The shape of the tissue in culture causes the hair follicles to grow into the dermal cysts, leaving them with nowhere to shed. Once researchers figure out how to allow the hair follicles to complete their natural cycle from the artificial environment of the culture medium, Koehler and his team believe the organoids could have important implications for toxicology and medicine. Moreover, Koehler thinks the mouse skin organoid technique could be used as a blueprint to generate human skin organoids.
"It could be potentially a superior model for testing drugs, or looking at things like the development of skin cancers, within an environment that's more representative of the in vivo microenvironment," says Koehler. "And it would allow us to limit the number of animals we use for research."
More information: Jiyoon Lee et al, Hair Follicle Development in Mouse Pluripotent Stem Cell-Derived Skin Organoids, Cell Reports (2018). DOI: 10.1016/j.celrep.2017.12.007