A look at the space between mouse brain cells
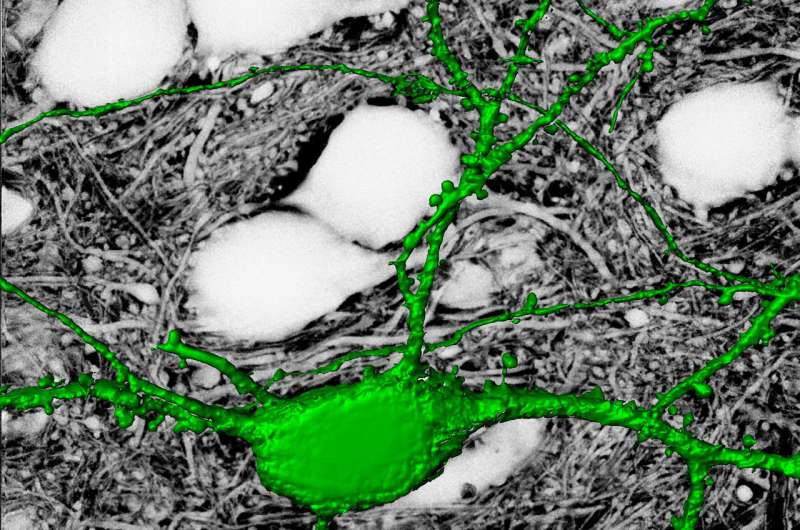
Between the brain's neurons and glial cells is a critical but understudied structure that's been called neuroscience's final frontier: the extracellular space. With a new imaging paradigm, scientists can now see into and study this complex fluid-filled matrix. The advance, demonstrated in mice, appears February 22 in the journal Cell.
The extracellular matrix—as much as 20 percent of the brain's overall volume—is "very convoluted, fascinating, and important," says senior author U. Valentin Nägerl of the University of Bordeaux's Institut Interdisciplinaire de Neurosciences. "These images give us a new sense of the raw and fluid complexity of brain tissue," he says.
To see it, Nägerl and colleagues coupled a diffusible, fluorescent dye with super-resolution microscopy, in this case stimulated emission depletion (STED) microscopy, to generate high-quality, 3D reconstructions of the tissue and spaces within brain slices kept alive outside the body.
The space between neurons is thought to regulate the flow of cerebrospinal fluid and clear metabolites during sleep, among other roles. Understanding its makeup could lead to better drug delivery within the brain. The extracellular space is altered during traumatic brain injury and epilepsy, yet its structure and function in healthy and diseased brains remains largely unknown.
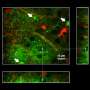
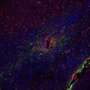
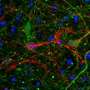
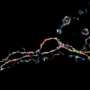
The technique revealed that the extracellular space is a profusion of creeping tendrils bursting from bulbous soma, or neuronal cell bodies, immersed in fluid. A green dye highlights a single neuron and its branching axons and dendrites that thin out as they wind like strands of Spanish moss around a million other axons.
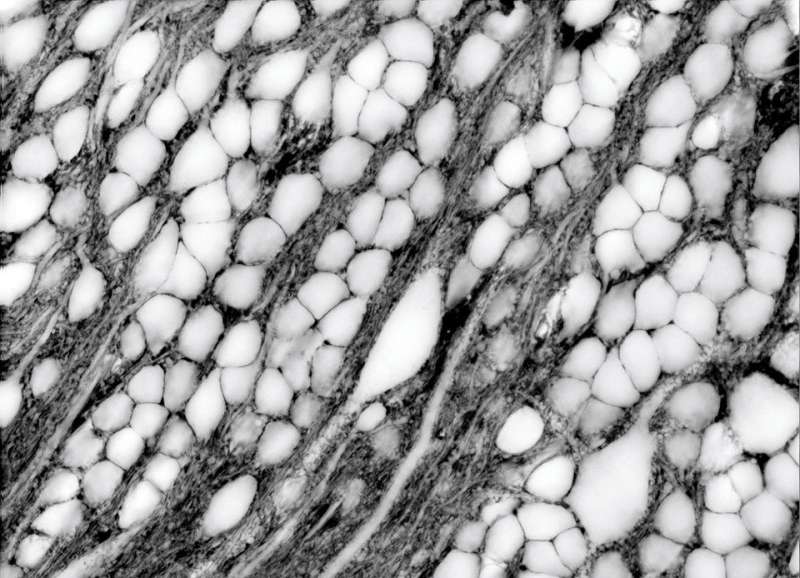
Because the extracellular space is essentially the negative imprint of the space taken up by cells, the technique depicts neurons and glia as sharp shadows, hence its name: super-resolution shadow imaging, or SUSHI. SUSHI's panoramic yet detailed views of the microanatomy of brain tissue is "like seeing the forest and the leaves at the same time," Nägerl says. And its ability to show the shape of dendrites and trace neuronal processes provides contextual information unavailable through imaging techniques such as electron microscopy, fluorescence microscopy, and magnetic resonance imaging (MRI).
SUSHI also visualizes synaptic clefts for the first time in slices of live tissue and reveals glia-neuron interactions and cell migration. As such, it can help researchers observe changes in synapses during brain development and disease. Advances in surgical techniques and adaptive optics may allow SUSHI to be used in the intact living brain in the near future, Nägerl says.
The extracellular space's fluid—a reservoir of ions critical for electrical activity and synaptic transmission—appears as tiny black spaces among white filaments and blobs. At one point, the researchers witnessed a microglial cell creeping, amoeba-like, through densely interwoven nerve fibers as it displaced a neuronal cell body and its bundle of axons. Nägerl was surprised by how variable and dynamic the extracellular space is, "which counters the impression we get from electron microscopy, where it looks super compact and rigid."
More information: Cell, Tønnesen et al. "Super-Resolution Imaging of the Extracellular Space in Living Brain Tissue." www.cell.com/cell/fulltext/S0092-8674(18)30151-X , DOI: 10.1016/j.cell.2018.02.007