New platform poised to be next generation of genetic medicines

A City of Hope scientist has discovered a gene-editing technology that could efficiently and accurately correct the genetic defects that underlie certain diseases, positioning the new tool as the basis for the next generation of genetic therapies.
This editing platform, discovered by City of Hope's Saswati Chatterjee, Ph.D., eventually may be used to cure inherited and acquired diseases.
"Our editing platform provides a new tool for the precise correction of genetic mutations in this rapidly growing field," said Chatterjee, senior author of the new study and a professor in the Department of Surgery at City of Hope. "Think of it as swapping out a mutated gene for a healthy gene to correct genetic mutations."
The proof-of-concept study, published in the journal Proceedings of the National Academy of Sciences on July 16, spotlights a promising new gene-editing platform that may eventually be used to treat diseases such as sickle cell disease, hemophilia (a condition that reduces the ability of blood to clot) and other genetic disorders, Chatterjee said.
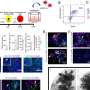
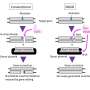
This genome-editing platform, tested using human blood and tissue as well as in preclinical models, is based on a family of nondisease-causing viruses known as adeno-associated viruses (AAV).
"Although injecting viruses into humans may sound alarming, a large portion of the population already has been exposed to AAV with no harmful consequences in their normal life," Chatterjee said.
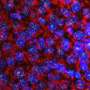
Chatterjee's research group isolated a subgroup of AAV known as AAVHSCs, which originate from human blood stem cells. The team discovered that AAVHSCs have the ability to efficiently deliver corrective DNA sequences to the nuclei of targeted cells in the body. Through a process called homologous recombination, these corrective sequences replace disease-causing genetic mutations in the genome. Since the therapeutic correction is at the genome level, it should lead to lifelong correction.
"We found that AAVHSC-based editing vectors can efficiently edit the genome following a single administration," Chatterjee said. "We hope to use these properties to develop widespread and accessible genome editing used to treat genetic diseases around the world."
The editing platform works efficiently in stem cells and mature cells, including adult liver and muscle cells. Successful utilization of AAV has the potential to change the world of gene editing, said Yuman Fong, M.D., co-author of the study and the Sangiacomo Family Chair in Surgical Oncology at City of Hope.
"We at City of Hope are attempting to build the foundation for another landmark treatment, like we did for synthetic human insulin," Fong said. "The potential of altering the course of genetic diseases is immense. Pairing the right AAV with blood stem cells is going to be an instrumental technique for precision medicine, the next frontier of medical treatment."
Chatterjee and her colleagues still have much work to do to characterize how this platform works and to develop it into therapeutics. They will address these questions in future studies.
This year, Chatterjee received a $2 million grant from the California Institute of Regenerative Medicine to develop a permanent cure for hemophilia A.
City of Hope licensed the pioneering AAV gene editing technology exclusively to Homology Medicines Inc. in May 2016. Chatterjee is the scientific co-founder of Homology Medicines and the chair of the company's scientific advisory board. The genetic medicines company went public in March 2018. Homology Medicines has entered into a research and development collaboration with Novartis.
More information: Laura J. Smith el al., "Stem cell-derived clade F AAV mediates high-efficiency homologous recombination-based genome editing," PNAS (2018). www.pnas.org/cgi/doi/10.1073/pnas.1802343115