How bone marrow regenerates after chemotherapy
Chemotherapy has a damaging effect on hematopoietic stem and progenitor cells (HSPCs) in bone marrow. However, once chemotherapy ends, HSPCs regenerate, a process that has remained unknown—until now. In a new study, researchers from Osaka University have identified the molecular mechanism by which HSPCs recover after injury.
HSPCs reside in the bone marrow and give rise to several types of blood cells, such as red blood cells (which carry oxygen), some white blood cells (which are important for the immune system) and platelets (which are necessary to stop bleeding). Because HSPCs constantly divide to generate new cells, they are particularly sensitive to injury induced by, for example, chemotherapy. Interestingly, HSPCs have the capability to regenerate upon injury.
"The bone marrow is a very active organ because it has to constantly produce new blood cells," says corresponding author of the study Masaru Ishii. "Once it loses its function, such as during chemotherapy, deadly conditions such as anemia, neutropenia and bleeding can occur. In this study, we wanted to understand how hematopoietic stem cells residing in the bone marrow regenerate upon chemotherapy-induced injury to recover their full function."
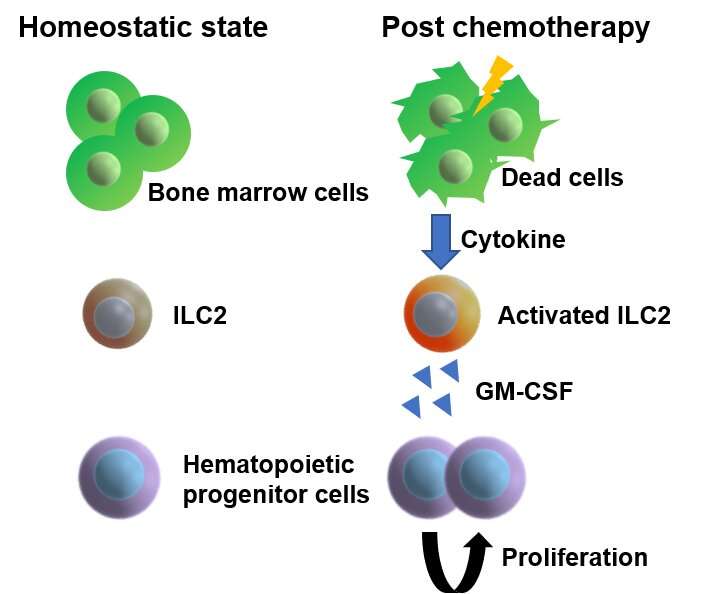
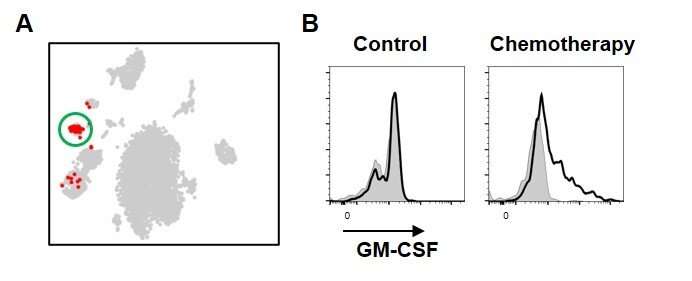
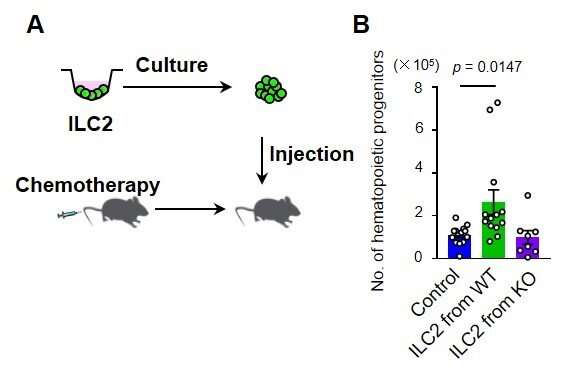
To achieve their goal, the researchers focused on a specific subset of blood cells that are produced from HSPCs, so-called group 2 innate lymphoid cells (ILC2s). While ILC2s exist in a number of tissues and play an important role in the immune system and tissue repair, those residing in bone marrow are thought to have a distinct role specific to their location. However, the nature of their function was unclear. To unravel the biological role of ILC2s, the researchers treated mice with 5-fluorouracil (5-FU), a chemotherapeutic agent toxic to HSPCs, and transplanted fresh, undamaged HSPCs into these mice, akin to a stem cell transplantation therapy in patients with leukemia. Interestingly, the researchers found that the injured HSPC microenvironment in 5-FU-treated mice promoted the proliferation of the transplanted HSPCs. By analyzing this finding at the molecular level, the researchers found that ILC2s in the bone marrow of treated mice produced granulocyte-macrophage colony-stimulating factor (GM-CSF) to aid in the process of HSPC regeneration.
But how exactly do ILC2s know that they should produce GM-CSF after bone marrow injury? To answer this question, the researchers widened their focus to investigate if there are other cells or molecules that direct ILC2s to the production of GM-CSF. They found that progenitors of antibody-producing B cells in the bone marrow produced interleukin (IL)-33 after injury, which in turn activated ILC2s, demonstrating how multiple molecular players are required to recover the damaged bone marrow. Importantly, the researchers showed that the transfer of isolated ILC2s to 5-FU-treated mice accelerates hematopoietic recovery, while the reduction of ILC2s results in the opposite effect, suggesting that ILC2s may serve as a sensor of bone marrow damage.
"These are striking results that show the bone marrow regenerates after chemotherapy," says first author of the study Takao Sudo. "Our results may contribute to the development of a novel therapeutic approach for chemotherapy-induced myelosuppression."
The article, "Group 2 innate lymphoid cells support hematopoietic recovery under stress conditions," was published in Journal of Experimental Medicine.
More information: Takao Sudo et al, , Journal of Experimental Medicine (2021). DOI: 10.1084/jem.20200817