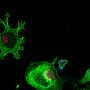
Sequential treatment with immunotherapy and checkpoint inhibitors prolongs anti-tumor activity
Advanced melanoma is one of the deadliest types of cancer, with a 5-year survival rate of only 27% for patients with distant metastases. Recent advances in targeted therapies and immunotherapies have greatly improved patient prognosis; however, many patients eventually develop resistance and disease recurrence. Researchers at Moffitt Cancer Center are investigating how to combine and sequence new therapies to improve survival. In a new article published in Cancer Immunology Research, the Moffitt team shows that sequential administration of immunotherapy followed by targeted therapy prolongs anti-tumor responses in preclinical models and may be a potential treatment option for patients.
One of the most common genetic alterations in melanoma are mutations of the BRAF gene, which affect approximately 50% of patients. These alterations result in downstream signaling through the protein MEK and stimulation of cell growth, invasion, and survival. Several therapies that target both BRAF or MEK have been developed and approved, and standard treatment for patients who have BRAF mutations is now combination therapy with BRAF and MEK inhibitors. Additionally, approximately 20% of melanomas have activating mutations in the NRAS gene, and there are no effective targeted therapies for this group of patients.
Immunotherapies have greatly improved patient outcomes by activating the immune system to target cancer cells, such as agents that target the proteins PD-1, PD-L1 and CTLA-4.
Given the success of both BRAF/MEK inhibitors and immunotherapies, researchers have attempted to combine these two classes of anti-cancer therapies. However, according to Keiran Smalley, Ph.D., director of Moffitt's Donald. A. Adam Melanoma and Skin Cancer Center of Excellence, "Initial attempts to develop targeted therapy/immunotherapy combinations clinically were not successful due to severe toxicity." Because of these toxicities, the Moffitt research team wanted to determine whether using a sequential treatment approach rather than a simultaneous combination approach would lead to durable anti-tumor activity in BRAF and NRAS-mutated melanoma.
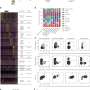
Researchers analyzed sequential combinations of immunotherapy followed by either BRAF/MEK targeted therapy or another targeted therapy combination (ceritinib/trametinib) and vice versa in preclinical mouse models. They discovered that immunotherapy followed by targeted therapy was more effective than either treatment approach alone. And while using targeted therapy followed by immunotherapy was better than targeted therapy alone, it was not as effective as the immunotherapy-targeted therapy sequence.
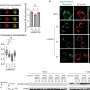
To determine why the immunotherapy-targeted therapy approach was effective, they performed laboratory experiments. They discovered the immunotherapy-targeted therapy sequence modulated the environment to promote immune cell functions resulting in anti-tumor activity while simultaneously blocking activity that permitted tumor cell escape from immune detection. The researchers also showed that the immunotherapy-targeted therapy sequence enriched the melanoma cells for proteins that promoted immune cell detection and suppressed signaling pathways associated with drug resistance.
The results from these studies suggest that sequential treatment approaches with anti-PD-1 agents followed by targeted therapy may be clinically beneficial, and several ongoing trials are currently investigating this hypothesis. "Together our data demonstrates that sustained anti-tumor responses to targeted therapy are dependent upon a vigorous, sustained immune response and that upfront use of immunotherapy can augment this," said Smalley.
More information: Manali S. Phadke et al, Targeted therapy given after anti-PD-1 leads to prolonged responses in mouse melanoma models through sustained antitumor immunity, Cancer Immunology Research (2021). DOI: 10.1158/2326-6066.CIR-20-0905