Immune cells promote proinflammatory fatty liver disease
A particular type of dendritic cell is responsible for the tissue damage that occurs in non-alcoholic steatohepatits (NASH) in mice and humans. The dendritic cells cause aggressive, proinflammatory behavior in T cells, as now discovered by researchers from the German Cancer Research Center (DKFZ) in collaboration with colleagues from Israeli research institutes. Blocking these dendritic cells alleviates symptoms in mice. This type of approach might also prevent the development of serious liver damage in NASH patients.
Obesity is extremely widespread in the Western world, and 90 percent of those affected show signs of fatty degeneration of the liver. If they maintain an unhealthy lifestyle over a long period (high-calorie diet, sedentary lifestyle), liver cell death occurs in around a fifth of these people, resulting in inflammation of the liver, referred to as non-alcoholic steatohepatitis (NASH). NASH can lead to liver fibrosis, life-threatening liver cirrhosis and liver cancer.
In addition to its well-known role in metabolism and in filtering toxins, the liver also has a strategic function as part of the immune system, acting as the primary line of defense against all microbial toxins and food contaminants that enter the body from the intestines via the portal vein. In order to perform this task, a whole army of different immune cells patrol the liver.
"We wanted to find out which immune or inflammatory cells in the liver promote NASH and the liver damage associated with it," explained Mathias Heikenwälder from the German Cancer Research Center (DKFZ). DKFZ researchers have now addressed the topic in collaboration with colleagues from the Weizmann Institute of Sciences and Sheba Medical Center in Israel. To do so, they analyzed the connection between the composition of the immune cell population in the liver and the degree of NASH-related liver damage. This enabled them to identify a particular type of immune cell that promotes progression of the disease—in both mice and humans.
Clue from laboratory mice on 'junk food'
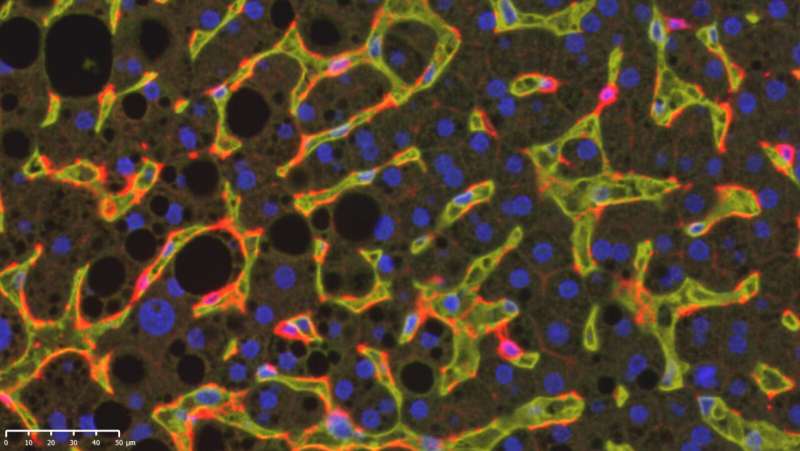
In order to investigate the immune system in NASH, the researchers fed laboratory mice a diet lacking essential nutrients but enriched with lipids and cholesterol—comparable to our 'junk food'—and observed the development of NASH. They studied the liver immune cells using single-cell RNA sequencing and discovered an unusually high number of a particular kind of cell, known as type 1 dendritic cells (cDC1), in the liver of NASH mice.
This phenomenon was not limited to mice. In tissue samples taken from patients in liver biopsies, the researchers found a correlation between the number of cDC1 cells and the extent of liver damage typical of NASH.
Do the cDC1 cells actually have an effect on liver pathology? The researchers pursued two channels of investigation here. They studied genetically modified mice lacking cDC1. In addition, they blocked cDC1 in the liver using specific antibodies. In both approaches, lower cDC1 activity was associated with a decrease in liver damage.
Dendritic cells normally only survive for a few days and need to be continually replaced by the immune system. The researchers discovered that the NASH-related tissue damage modulates the hematopoietic system in the bone marrow, as a result of which the cDC1 precursors divide more often and replenish the supply more readily.
Dendritic cells induce aggressive behavior in T cells
In a normal immune response, dendritic cells screen the organs for conspicuous immunologic features and then continue on to the neighboring lymph nodes—the command centers of the immune response—to pass on this information to the T cells. In NASH subjects, the German-Israeli team has now discovered that the cDC1 induce inflammatory and more aggressive behavior in T cells in the lymph nodes responsible for the liver, causing liver damage and leading to progression of the disease. "It is only recently that we identified these autoaggressive T cells as being responsible for liver damage in NASH. Now we also understand what induces this harmful behavior," Mathias Heikenwälder remarked.
Now that the cDC1 have been shown to play a key role in the progression of NASH, targeted manipulation of these cells might offer a new way of treating inflammation of the liver and its serious repercussions. "We are increasingly recognizing that certain cells of the immune system are involved in the development of different diseases, including cancer, diabetes, and Alzheimer's disease. Medicine is thus increasingly using ways of modulating the immune system and using drugs to push it in the right direction. This kind of approach might also work to prevent serious liver damage in NASH patients," Heikenwälder explained.
Eran Elinav, also a senior author of the study and head of research groups at DKFZ and the Weizmann Institute, believes that it is highly probable that gut bacteria affect the immune cells in this disease: "We now aim to find out how the gut and its bacteria influence activation of the immune cells in the liver. By doing so, we hope to be able to develop new treatment strategies."
The study is published in Nature Medicine.
More information: Deczkowska, A., David, E., Ramadori, P. et al. XCR1+ type 1 conventional dendritic cells drive liver pathology in non-alcoholic steatohepatitis. Nat Med (2021). doi.org/10.1038/s41591-021-01344-3 , www.nature.com/articles/s41591-021-01344-3#citeas