Treating orphan diseases in children
Blau syndrome is an extremely rare autoimmune disease estimated to occur in fewer than one in a million people. Patients begin to show symptoms before the age of 4, and the symptoms will progress to cause serious complications such as blindness and joint immobility. A new study by CiRA scientists discovers that immune cells in patients are primed to exert an immunological response and how certain treatments can prevent this from happening.
With fewer than 100 recorded patients in Japan, CiRA Associate Professor and pediatrician Megumu Saito said there is little clinicians can do to help Blau syndrome patients.
"In the past, the only treatment option was steroids, but they only treat the symptoms, and the patients' conditions will continue to worsen. Recently, anti-cytokine therapies, especially anti-TNF therapy have been used, but we do not understand how they work."
Dr. Yohko Kitagawa, the first author of the study, noted that TNF is core to the autoimmune reaction.
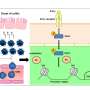
"In Blau syndrome, macrophages excessively produce TNF even in the absence of pathogens, stimulating more IFNγ from lymphocytes, which further activates the macrophages. It becomes a dangerous positive feedback," she said.
The immune system includes a number of cell types including macrophages and lymphocytes. These cells each serve their own purpose, but like the electrician, plumber and carpenter, they also depend on each other. In response to pathogens, macrophages communicate with lymphocytes by secreting TNF, while lymphocytes respond by secreting IFNγ. Both these cytokines are fundamental to the immune system.
The study shows that macrophages from untreated patients showed a different pattern of gene expressions than those from healthy donors when they were stimulated with IFNγ. Kitagawa said that this difference indicated that the macrophages were in a condition primed for a strong immune response.
On the other hand, macrophages from patients treated with anti-TNF agents did not show the abnormal cytokine production and instead behaved like macrophages from healthy donors.
However, one concern about using patient immune cells, is that a treatment itself can affect the cells, affecting experimental results. Therefore, the researchers also prepared macrophages from patient iPS cells.
"We wanted to distinguish environmental effects caused by the treatment from the actual disease condition of the macrophages. The anti-TNF treatment could affect the state of the macrophages, but it would not have this effect on iPS cells," said Saito.
Genetically, Blau syndrome is the result of a mutation in the NOD2 gene, which codes for an important immune protein. Patient iPS cells were thus prepared, and some were genetically edited to correct the NOD2 mutation. All were then differentiated into macrophages. These macrophages were stimulated with IFNγ and found to behave like macrophages taken directly from the patient.
"The NOD2 mutations caused an accelerated inflammatory response upon IFNγ stimulation. Our findings suggest the patient cells are in a primed state that have a lower threshold for activation. The anti-TNF treatment raises this threshold to prevent the pathological loop between macrophages and lymphocytes," says Kitagawa.
More information: Yohko Kitagawa et al, Anti-TNF treatment corrects IFN-γ–dependent proinflammatory signatures in Blau syndrome patient–derived macrophages, Journal of Allergy and Clinical Immunology (2021). DOI: 10.1016/j.jaci.2021.05.030