Targeted tumors attack not-innocent bystanders
How do you kill tumor cells that can't be targeted? Get their more susceptible neighbors to help.
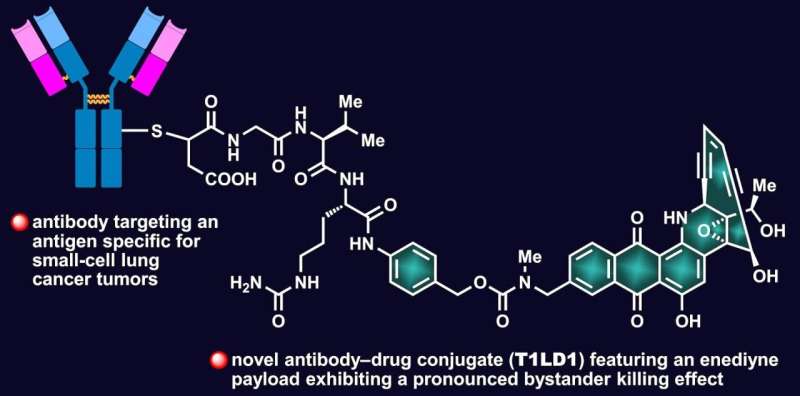
The Rice University lab of synthetic chemist K.C. Nicolaou, in collaboration with AbbVie Inc., has created unique antibody-drug conjugates (ADCs) that link a synthetic uncialamycin analog to antibodies that target cancer cells.
Once they enter the targeted tumor cells, these ADCs exhibit a "significant bystander effect," according to the study. In other words, cancerous neighbor cells that aren't directly attacked by the drugs are also affected.
The study in the Proceedings of the National Academy of Sciences presents "an intriguing opportunity in the tumor-targeting therapeutics development area," according to the researchers.
"The bystander effect is expected to improve the efficacy of the ADC drug by killing, in addition to the targeted cancer cells, a significant number of nearby, nontargeted cancer cells usually found within or in close proximity to malignant tumors," Nicolaou said.
Nicolaou's lab, which specializes in the synthesis of molecules found in nature that have medicinal properties, first reported in 2016 its development of a method to simplify its synthesis of uncialamycin, an enediyne compound isolated from a strain of marine bacteria. Once inside the nucleus of a cell, it produces radicals that cut both strands of the target's DNA.
The earlier study produced 13 analogs of uncialamycin and tested them on lung, gastric, ovarian and multidrug-resistant cancer cell lines with "remarkably high potency," the researchers reported at the time, suggested they showed promise as ADC payloads and chose one for further development.
That promise is now paying off. In tests on solid tumors, the best of a variety of ADCs killed not only the cell that expressed the target protein but also 'neighbor' cancer cells that didn't.
"That is unique within the enediyne payload-carrying ADCs, and in contrast to the currently employed ADCs containing N-acetyl calicheamicin payloads, which do not exhibit this killing effect," Nicolaou said.
Importantly, he said, the majority of clinically approved ADCs demonstrate bystander killing effect.
The study found ADCs made with cleavable linkers that are stable in the bloodstream but release their payloads in the tumor microenvironment were most effective at decreasing the size of small-cell lung cancer tumors in PDX mouse models.
More information: K. C. Nicolaou et al, Uncialamycin-based antibody–drug conjugates: Unique enediyne ADCs exhibiting bystander killing effect, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2107042118