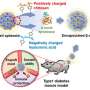
Building a better bioartificial pancreas
More than 40 million people worldwide are affected with type 1 diabetes (T1D) mellitus, an autoimmune disease in which insulin producing β-cells in the pancreas are destroyed by the immune system. Today, there are several new and emerging treatment methods for type 1 diabetes, including macroencapsulation devices (MEDs)—compartments designed to house and protect insulin-secreting cells. Like a suit of armor around a knight, the MEDs protect the cells within it from attack (from the host immune system) while allowing nutrients in and out so that the cells can continue to survive. But MEDs have several limitations and scaling up such devices for use in humans has been challenging. A team of researchers from Brigham and Women's Hospital in collaboration with colleagues at Harvard University and the University of Massachusetts Medical School has designed a convection-enhanced MED (ceMED), which can continuously bathe cells in the nutrients they need and improve cell loading capacity, while increasing cell survival, glucose sensitivity and timely insulin secretion. In preclinical models, the ceMED rapidly responded to blood sugar levels within two days of being implanted. Results are published in The Proceedings of the National Academy of Sciences.
"Thanks to recent advances, we're getting closer and closer to having an unlimited source of β-like cells that can respond to glucose by secreting insulin, but the next challenge is getting those cells into the body in a way that's minimally invasive and will have longevity with maximal function," said corresponding author Jeff Karp, Ph.D., principal investigator and Distinguished Chair in Clinical Anesthesiology, Perioperative and Pain Medicine. "Our device demonstrated enhanced cell viability and minimal delay following transplantation. It's a strong preclinical proof of concept for this system."
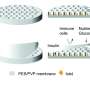
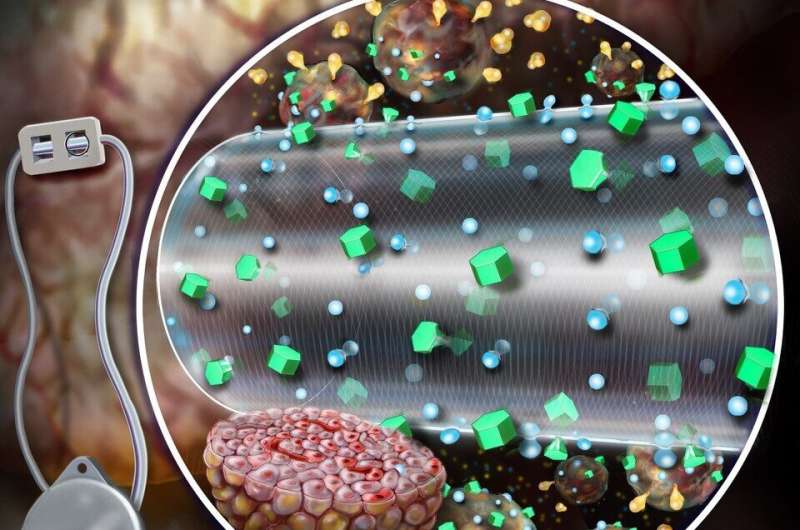
Current MEDs are diffusion dependent—nutrients diffuse across the outer membrane of the device and only a number of cells may receive nutrients and oxygen and, in turn, secrete insulin. The ceMED was designed to provide convective nutrients through a continuous flow of fluid to the encapsulated cells, allowing multiple layers of cells to grow and survive. The team's prototype features two chambers—an equilibrium chamber (EqC) that collects nutrients from the surroundings and a cell chamber (CC) that houses the protected cells. The EqC is enclosed in polytetrafluoroethylene—a semi-permeable membrane with pores that allow fluids in. An additional inner membrane surrounding the CC selectively allows for nutrient transport and protects against immune responses. Perfused liquids flow through a porous hollow fiber reaching the CC at a similar concentration of nutrients as the tissue surrounding the implant. The hollow fiber allows insulin and glucose to freely pass but does not allow key immune molecules in that could attack the encapsulated cells.
"The application of stem cell-derived islets to treat autoimmune or Type 1 diabetes has now moved to the point of finding a method to protect the cells from immune rejection and maximizing their survival and function following transplantation," said co-author Doug Melton, Ph.D., of the Department of Stem Cell and Regenerative Biology at the Harvard Stem Cell Institute. "Convection-enhanced macroencapsulation may well be a viable approach to achieve all of these goals."
The device offers many advantages over conventional insulin pumps and allows cells to secrete insulin on demand and quickly stop secreting insulin as blood glucose levels decline. In rodent models of type 1 diabetes, the ceMED enhanced the survival and insulin secretions of cells and began to decrease blood glucose level as early as two days post-transplantation.
"The ceMED device has the potential to be an autonomous system that would not require constant refilling and replacement of insulin cartridges," said lead author Kisuk Yang, Ph.D., a former postdoctoral fellow in the Karp Lab and now faculty at the Division of Bioengineering at Incheon National University in South Korea.
"Due to its responsiveness, this device and novel flow-enhanced approach could be particularly useful for 'brittle' diabetics, people whose diabetes results in unpredictable swings in blood sugar levels," added Eoin O'Cearbhaill, Ph.D. (now at University College Dublin, Ireland), a co-author who helped develop this concept while working as a postdoctoral fellow in the Karp Lab. The team notes future directions that will need to be pursued to bring the device to the clinic, including scaling up cell loading capacity and optimizing the perfused flow system for human use.
"Overall, these results highlight significant advantages of ceMED over existing diffusion-based devices including improved cell survival, reduced fibrous encapsulation that can compromise functionality over time, and quicker on and off rates for insulin secretion" said Karp. "This approach has the potential to enhance the success of β cell replacement therapies to help many T1D patients and their families manage this challenging disease."
More information: A therapeutic convection–enhanced macroencapsulation device for enhancing β cell viability and insulin secretion, Proceedings of the National Academy of Sciences (2021). DOI: 10.1073/pnas.2101258118