This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Wt1 gene found to be a key factor in the endothelial cell during the formation of new coronary vessels
How is the mammals' heart formed? What role do endothelial cells play? Do the same pathways and genes involved in the formation of the embryonic heart also play a role in repair processes? A new study conducted by a team from the Faculty of Biology and the Institute of Biomedicine of the UB (IBUB) reveals that the deletion of the Wt1 gene in endothelial cells during the early stages of coronary vessel formation affects the correct development of the heart in embryonic stages.
The study, featured in the Research Highlight section of the journal Development, is led by Professor Ofelia Martínez-Estrada, from the Department of Cell Biology, Immunology and Physiology of the Faculty of Biology, member of the IBUB and the Celltec UB research group.
The article, whose first author is the researcher Marina Ramiro-Pareta (UB-IBUB), also involves teams from the Faculty of Medicine and Health Sciences and the UB Institute of Neurosciences (UBNeuro), the Germans Trias i Pujol Research Institute (IGTP), the Autonomous University of Barcelona (UAB), the National Centre for Genomic Analysis (CNAG-CRG) and the Centre for Biomedical Research Network on Cardiovascular Diseases (CIBERCV).
New perspectives on Wt1 gene functions
Endothelial cells form the inner cell layer of blood vessels and have a wide range of essential functions in the development of tissues and organs such as the heart, a highly vascularized organ. In the cardiovascular system, the Wt1 gene is highly expressed in the epicardium—the outer layer of the heart—and in the endothelial cells of this organ.
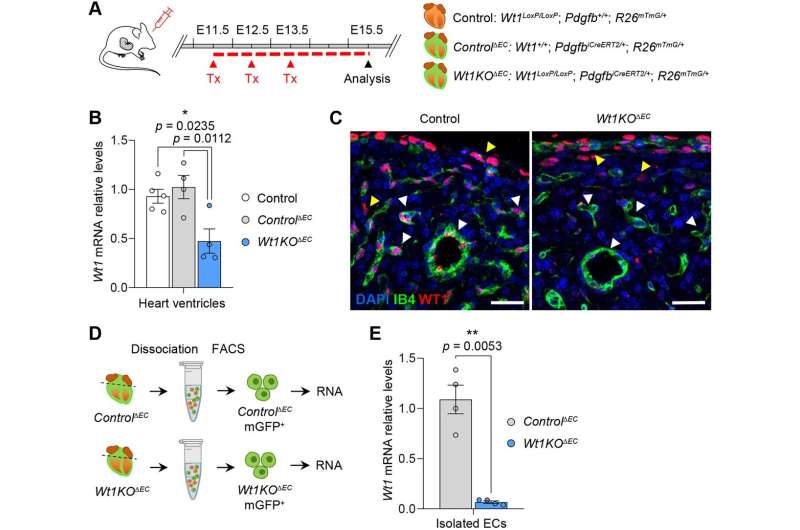
"To date, it was thought that the defects in embryonic heart formation observed in Wt1KO transgenic mouse models—i.e. mice lacking this gene—were mainly due to the functions of this gene in the epicardium," notes Professor Ofelia Martínez-Estrada. "Now, our study shows that deletion of the Wt1 gene in the endothelial cell affects coronary vessel formation and myocardial development."
In this study, carried out on animal models, the researchers combined image analysis and RNA-Seq techniques to identify how the deletion of Wt1 in coronary endothelial cells during the early stages of development modifies the transcriptional signature—i.e. the different gene expressions—of these cell types.
"In particular, many of the genes found to be modified in these Wt1KO cells are genes that are dynamically modulated throughout the development of coronary endothelial cells. These transcriptional changes correlate with defects in the formation and differentiation of arteries and veins, as well as with a decrease in cardiomyocyte proliferation, two processes that are crucial for the correct development of the embryonic heart," states researcher Marina Ramiro-Pareta.
Revascularizing the heart after a heart attack
The formation of new blood vessels after a myocardial infarction is essential for the survival of the damaged tissue. Although various therapeutic strategies aimed at stimulating myocardial angiogenesis after a cardiac infarction have been adopted in recent years, there are still no effective therapies to modulate the process of de novo vascularization, which is insufficient in mammals.
"The results of the new study have a number of important implications for the field of cardiac development. They also confirm the need for further research into the role of the Wt1 gene in endothelial cells in the revascularization processes of ischemic hearts," concludes Professor Ofelia Martínez-Estrada.
More information: Marina Ramiro-Pareta et al, Endothelial deletion of Wt1 disrupts coronary angiogenesis and myocardium development, Development (2023). DOI: 10.1242/dev.201147