This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Mini livers recreate real environment for drug discovery
Fatty liver disease is exploding globally, affecting as many as 30% of people in Western countries. Apart from alcohol, obesity and diabetes are among the most dominant risk factors.
"There is a huge focus on seeing if there is anything you can do to inhibit this explosion of fatty liver disease medically," says Professor Niels Bent Larsen from DTU Health Tech.
Along with colleagues from Norway, Sweden, Germany, and Czech Republic his team at DTU Health Tech have developed a new technology that may speed up the process of finding new drugs to combat liver diseases. Their results have just been published in the journal Acta Biomaterialia.
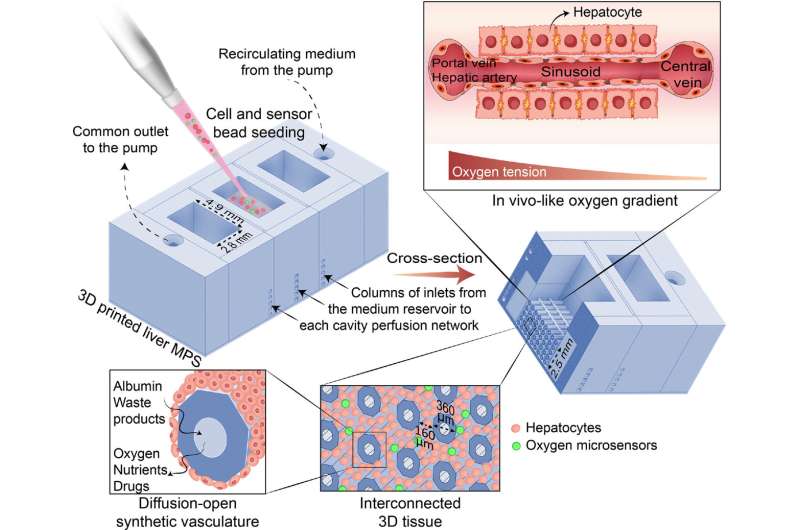
"To find new drugs efficiently, you need models, and you want them to mimic the real thing—its structure and function—as closely as possible. So, what we've done in our labs is to make a real mini liver. Basically, we 3D print a bundle of artificial blood vessels and pour live liver cells in between them."
"Because the sides of these artificial blood vessel are very close together, we can supply the liver cells with the same conditions they have inside our liver. By sending in liquid with nourishment and oxygen through the artificial blood vessels we can keep them running for months as a fused liver-like tissue with retained liver functions."
Model recreates real environment
The human liver is made of tiny units called acini, which have zones that do different things and need different amounts of oxygen and nutrition. The zone closest to the blood supply will receive the most oxygen, which supports oxidative energy metabolism and glucose synthesis crucial to studying metabolic disease.
The liver cells consume the oxygen in the blood flowing through the acinus, so the zone furthest from the blood supply receives much less oxygen. This is the home of enzymatic processes that convert toxins and drugs arriving from the small intestine into safer compounds, but also where toxic byproducts can kill the liver cells leading to permanent liver damage.
Current methods for modeling the liver are prone to losing their features quickly. Also, they do not have zones with different oxygen levels and functions, which you need if you want to realistically test how the liver responds to different drugs.
The researchers tested how the common pain killer paracetamol affected the cells at different oxygen levels and saw that the cells in the low-oxygen zone were more sensitive to the drug. Since paracetamol is only processed in this zone it is a good indicator that you have a viable liver model. They also looked at the proteins made by the cells and saw that their distribution between zones matched that of human liver acini.
"To my knowledge, our model is the only one out there that can recreate this real environment only from having the liver cells themselves produce the natural gradual change in oxygen and metabolites through the acinus. I believe it shows great promise that we have been able to make real, living tissue that we can monitor and experiment on in 3D and in an environment that closely mimics the real liver. This gives us a huge amount of control and will enable drug developers to speed up their testing significantly," says Niels Bent Larsen.
More information: Milan Finn Wesseler et al, 3D microperfusion of mesoscale human microphysiological liver models improves functionality and recapitulates hepatic zonation, Acta Biomaterialia (2023). DOI: 10.1016/j.actbio.2023.09.022