This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Researcher discusses turbocharging CRISPR to understand how the immune system fights cancer
Over the past two decades, the immune system has attracted increasing attention for its role in fighting cancer. As researchers have learned more and more about the cancer-immune system interplay, several antitumor immunotherapies have become FDA-approved and are now regularly used to treat multiple cancer types.
Yet despite these advances, much remains unknown about how the immune system fights cancer—and about immunity in general, said Martin LaFleur, a postdoctoral fellow in the laboratory of Arlene Sharpe, chair of the Department of Immunology in the Blavatnik Institute at Harvard Medical School.
CRISPR-based gene editing, in which scientists modify the genome using a tool developed just over a decade ago, has become a mainstay of biological discovery, providing relatively quick insight into the function of individual genes and targets for new therapies.
However, LaFleur said, this approach is not without challenges. Chief among them is that it is hard to modify immune cells without changing their biology, which hampers the ability to study immune cell behavior in its full complexity in a living organism.
Now, LaFleur, Sharpe, and their team have succeeded in bypassing this hurdle by deploying CRISPR in a new way to study the function of immune genes.
Their work, described in two papers—one in Nature Immunology and the other in the Journal of Experimental Medicine—could eventually yield insights about cancer immunology as well as about other diseases driven by immune system dysfunction.
Harvard Medicine News spoke with LaFleur about what this advance means for the future of immunology research.
Let's set the stage with a refresher on how CRISPR works
Programmable CRISPR-based gene editing was developed in 2012 and became such a powerful tool for biologic research that its discoverers won the Nobel Prize in Chemistry in 2020.
The CRISPR gene-editing system uses an enzyme called Cas-9 that acts like a pair of molecular scissors that cuts both strands of DNA and in doing so disrupts or knocks out the function of a gene. To select the gene to knock out, this system uses a complementary piece of RNA that matches the gene and acts as a guide. It's a very flexible approach for very quickly knocking out and studying the function of almost any gene you want.
How is CRISPR used to understand the immune function of genes?
Immune cells interact with a lot of other cell types that can't be modeled well in petri dishes, so we prefer immune studies to happen inside a living organism like a mouse—a far more reliable way to capture the complexity of cell-to-cell interactions as they occur in the body rather than in a lab dish. CRISPR editing inside the body is difficult, so immune cells typically need to come out and be modified using this tool in a petri dish. The edited cells are then put back into the body.
However, only certain immune cell types can be incorporated efficiently when transferred back into a mouse. Also, the actual process of manipulating immune cells in a dish can change their biology, so you may not be studying what you actually want to study once they're removed from the body.
Also, CRISPR had been used only to turn off a single gene at a single time in immune cells. But our cells contain thousands of genes, so what if we want to knock out multiple genes in different cell types at different times in the same animal? This would provide greater insight into the complexities of genes and their interactions in immune cells over time.
How does your new study address these challenges?
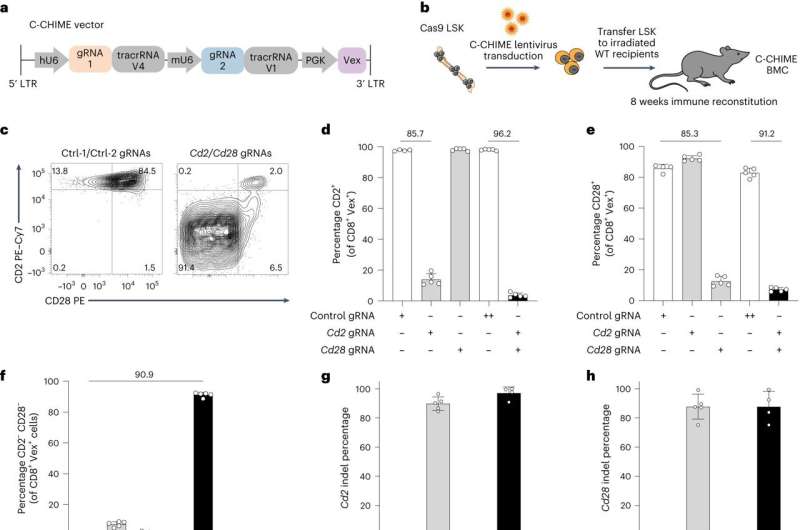
We decided to take a completely different approach for using CRISPR. Rather than directly modify the immune cells we're interested in, we modified their precursors, the stem cells found in bone marrow that produce all immune cells. We removed those from mice and used CRISPR to knock out the genes we were interested in, and then replaced these stem cells in mice whose native bone marrow stem cells had been removed. We call this system CHimeric IMmune Editing, or CHIME.
In an earlier study, we used CHIME to knock out a gene called Ptpn2, which has shown some promise for cancer immunotherapy, one of the focuses of the Sharpe Lab. When we deleted that one gene in a subset of immune cells known as CD8+ T cells, they became better cancer fighters.
With our Nature Immunology study, we wanted to see if we could modify CHIME and make it both more precise and more versatile. We used it to knock out two genes at once in several different cell types; we deployed it to target genes specifically in a single cell type; we used CRISPR to disrupt genes in modified cells once they were already back inside the animal; and we also used it to knock out two different genes at different points in time.
We used different tactics, such as packaging multiple guide RNAs together and using a trick that disables genes only under certain circumstances, such as when mice receive a drug. We were able to demonstrate that each of these strategies is feasible.
What is your ultimate goal with this research?
Our ultimate goal is to better understand the immune system, particularly in its capacity to fight cancer. We want to encourage strong anticancer immunity—meaning we want to optimize how immune cells fight tumors—but also want spare healthy cells and tissues from the immune attack. This requires a very nuanced calibration of the immune system and can be a tricky balance.
Moreover, the benefits could extend beyond cancer and be applied to many other diseases driven by the immune system, including autoimmune conditions.
What are your next steps?
We just published a second paper [in the Journal of Experimental Medicine] that lays out a framework for the field in studies using CRISPR to screen immune gene function in living animals. Central to our framework is adding a genetic "barcode" to CRISPR-edited immune cells so we can track them as they multiply and spread within animals.
We're hoping that this framework and CHIME will give researchers new tools to study immune cells in cancer or any other disease model of their choice, eventually leading to new immune-centered therapies.
More information: Martin W. LaFleur et al, X-CHIME enables combinatorial, inducible, lineage-specific and sequential knockout of genes in the immune system, Nature Immunology (2023). DOI: 10.1038/s41590-023-01689-6
Lauren E. Milling et al, Framework for in vivo T cell screens, Journal of Experimental Medicine (2024). DOI: 10.1084/jem.20230699