Immunotherapy approach to Alzheimer's studied in fly models
At Genetics Society of America's Drosophila Research Conference, scientists will report on results of using fly models to investigate passive immunotherapy to block amyloid-β42 peptides of amyloid plaques that damage the brain cells of patients with Alzheimer's.
Developing treatments that slow, if not halt, the neuronal loss and cognitive decline of Alzheimer's disease (AD) has proven to be a challenge. Among the scientists who have taken on that challenge are researchers at the University of Florida's McKnight Brain Institute in Gainesville, using the fruit fly Drosophila melanogaster as a model.
The scientists are investigating passive immunotherapy, one of the most promising approaches to blocking the amyloid-β42 (Aβ42) peptide, the main component of the amyloid plaques that damage the brain cells of patients with AD.
Monoclonal antibodies, the cornerstone of immunotherapy, do not easily penetrate the tightly packed cells of the blood–brain barrier. As alternatives to full antibodies, the University of Florida scientists generated transgenic flies expressing two anti-Aβ single chain variable fragments (scFv), known as scFv9 and scFv42.2.
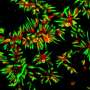
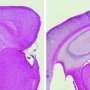
Drosophila flies genetically modified to express human Aβ42 also co-expressed either scFv9 or scFv42.2. Each treatment reduced the loss of photoreceptor neurons due to Aβ42 toxicity and improved the insects' eye morphology. In addition, the flies' mushroom bodies, the brain structures involved in olfactory learning and memory, were protected.
Administering both scFv9 and scFv42.2 to the flies produced a synergistic protective activity, which suggests that both scFvs can simultaneously target different Aβ42 epitopes, the scientists said. This is the first time that two antibodies targeting different regions of Aβ42 have shown synergistic activities, thanks to the experimental flexibility of Drosophila.
In addition to immunotherapy, the scientists investigated the protective effects of a laboratory engineered secreted Heat shock protein 70 (secHsp70) on flies whose brain cells were accumulating Aβ42 extracellularly. Hsp70 is a chaperone that prevents and reverts protein misfolding but is primarily located inside the cell. In flies treated with extracellular secHsp70, the eyes were perfectly organized, cell death was reduced, calyces in the mushroom bodies were preserved, and locomotor function was improved.
The researchers determined that secHsp70 interacted directly with Aβ42 without affecting Aβ42 accumulation and aggregation in the flies' brains. Thus, the protective activity of secHsp70 may be mediated by stable binding to toxic Aβ42 species. This study is a new approach to broadly target amyloids outside the cell and block their ability to attack the surrounding neurons, the scientists said.
More information: Abstract: "Exploiting Drosophila as a platform to develop anti-amyloid strategies." Pedro Fernandez-Funez1,2, Jonantan Sanchez-Garcia1, Swati Khare1, Alfonso Martin-Peña1, Diego Rincon-Limas1,2. 1) Dept Neurology, Univ Florida, Gainesville, FL; 2) McKnight Brain Institute, Genetics Institute, and Center for Translational Research on Neurodegenerative Disorders. abstracts.genetics-gsa.org/cgi … il.pl?absno=14531635