Research team describes novel tumor treatment
In the first published results from a $386,000 National Cancer Institute grant awarded earlier this year, a paper by Scott Verbridge and Rafael Davalos in Scientific Reports has been published.
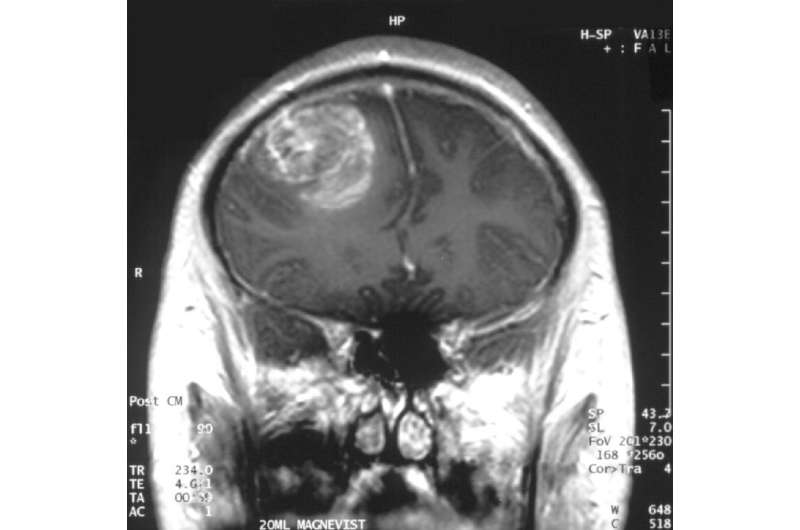
The paper—co-written by John Rossmeisl , Virginia-Maryland College of Veterinary Medicine associate professor of internal medicine and neurology; Michael Sano, postdoctoral researcher at Stanford; and Virginia Tech Department of Biomedical Engineering and Mechanics Ph.D. students Jill Ivey of Fayetteville, Arkansas and Eduardo Latouche of Valencia, Venezuela,—describes the researchers' work on developing a new type of treatment for glioblastoma multiforme (GBM), the most common and deadly malignant primary brain tumor.
Patients with glioblastoma have a five-year survival rate of less than 10 percent, Verbridge said.
"This statistic has not improved significantly in decades, and there is still no treatment option to preferentially target the glioma stem cells or diffuse infiltrative cells that lead to tumor recurrence after surgery, chemo, or radiotherapy," Verbridge said.
The paper, "Targeted cellular ablation based on the morphology of malignant cells," describes research into a new treatment option involving pulsed electric fields (PEFs) that is better at targeting and killing malignant cells while leaving healthy cells alive.
Glioblastoma is particularly resistant to conventional and targeted cancer therapies, in part because tumors are made up of cells that are very different from one another. Surgical, chemical, radiation, and molecular treatments all have shortcomings that make it difficult to target malignant cells without damaging critical surrounding structures or prompting the emergence of therapy-resistant cells.
Recurrence after initial treatment is extremely common as a result.
In their work, Verbridge and Davalos are building on a technique patented by Davalos involving irreversible electroporation (IRE) of tumor cells by inserting electrodes directly into a tumor. This treatment has been successfully used to treat canine patients with similar brain tumors.
Verbridge and Davalos have developed a high-frequency version of this therapy (HFIRE) that uses pulses delivered in a rapid burst. IRE and HFIRE are both capable of killing tumor cells, but the mechanism differs, the researchers found. IRE appears to cause cellular death by disrupting the outer cell membrane. HFIRE, in contrast, appears to also cause nuclear collapse. Mathematical models developed by the researchers predicted that the nuclear damage should scale with size of the nucleus relative to the cell.
Because HFIRE targets the nucleus, the researchers hypothesized that malignant cells, which have larger nuclei compared to normal cells in many cancers including glioblastoma, would have a lower threshold for a lethal burst.
Therefore, properly tuned HFIRE could target malignant cells while leaving normal, healthy cells unharmed.
This was confirmed using 3-D tissue models consisting of co-cultures of normal and malignant brain cells. "We're essentially destroying tumor cells based on their altered morphologies, specifically their enlarged nucleus-to-cytoplasm ratio which has been known for decades to be a strong indicator of malignancy in many cancers. This is a completely different potential approach to targeted therapy," Verbridge said.
Future experiments in vivo will be necessary to determine exactly how selective HFIRE ablation therapy will be, but the researchers expressed confidence that such a therapy, either on its own or in combination with other treatments, could be effective at killing the invasive glioblastoma cells that so often lead to tumor recurrence after initial rounds of treatment.
More information: Targeted cellular ablation based on the morphology of malignant cells, Scientific Reports, 2015.