Researchers suppress fibrosis chemical signal to block haywire healing
An injured body always seeks to heal. But that process is far from simple. A host of cells organize to restore what was damaged. Then, critically, the process tapers off. And when it doesn't, the effects can be disastrous. Fibrosis is the thickening and scarring of tissue due to an overactive healing response.
"It has been reported that upward of 45 percent of all deaths in the developed world can be attributed to a disorder with a link to fibrosis," says Edward Leof, Ph.D., senior author on a new paper in the Journal of Clinical Investigation and co-leader of the Immunity and Fibrosis platform within Mayo Clinic's Center for Biomedical Discovery. "It can take place in pretty much any tissue or organ in the body."
In the intestines, fibrosis can be linked to inflammatory bowel disease. It can damage the liver as it heals after viral infections or excessive alcohol consumption. Glaucoma and macular degeneration are forms of fibrosis in the eyes. And, in the lungs, fibrosis plays parts in asthma and chronic obstructive pulmonary disease. The process of fibrosis fills arteries with fibrous plugs (atherosclerosis). It also allows scar tissue to creep over healthy skin in a disease called keloid disorder.
Targeting TGF-beta
TGF-beta is a growth factor important for a variety of developmental and cellular activities. It is the most pro-fibrotic cell signaling molecule known. TGF-beta is referred to as a master switch in the genesis of organ fibrosis, released by many cells at the point of injury. Dr. Leof and team identified a new approach to modulating the fibrosis-related actions of TGF-beta by blocking a key protein.
Tagalong protein derails TGF-beta
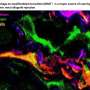
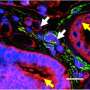
The key protein is called SMAD3. The SMAD proteins work within cells to read the messages transmitted cell-to-cell, and respond by activating gene transcription. In a complex with other SMAD proteins and a protein called SNX9, SMAD3 crosses into the nucleus of the cell and starts interacting with genes to promote pro-fibrotic TGF-beta signaling.
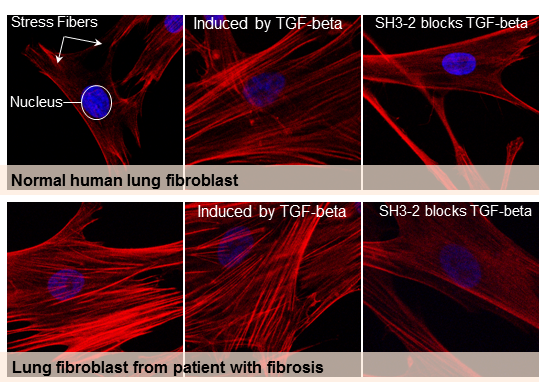
To block SMAD3 and prevent this TGF-beta response, Dr. Leof's team fused a snippet of the SNX9 protein with a short chain of amino acids specifically designed to carry a protein cargo into a cell. When the snippet of SNX9 binds to SMAD3, that complex prevents SMAD3 from entering the nucleus and impacting (activating and/or suppressing) genes regulated by TGF-beta.
Where the peptide-carrying SNX9 interacted with SMAD3, inhibition of TGF-beta activity was clear. The authors tested the effect in human and mouse cell cultures, as well as in two animal models of lung fibrosis.
New data, new drug theories
Because of the complexity of TGF-beta—that it both stimulates and suppresses genes regulating a number of important physiological processes—drugs that target all TGF-beta actions generally have unacceptable side effects. As such, there is no effective treatment for the process of fibrosis.
"This paper provides proof of concept that by targeting specific signaling molecules you can show efficacy in vitro and in vivo," explains Dr. Leof. "This may be a way to get rid of the bad actions but maintain other good things TGF-beta does."