Team devises a new method that could speed up vaccine development for HIV
Around 1% of patients with HIV - known as elite controllers - are able to survive without antiviral treatment, because their immune systems produce certain kinds of HIV-specific antibodies: proteins that recognise features on the surface of the virus and bind to them, making the virus inactive. The challenge in developing an HIV vaccine is to identify specific features in the proteins on the virus's surface which are recognised by the immune system and elicit a response similar to that seen in elite controllers.
A widely used technique for studying proteins on the surfaces of cells - which is sometimes also used with viruses - is fluorescence-activated cell sorting (FACS). You take a sample of cells and add fluorescent antibodies that bind to the surface proteins you're interested in. Cells with proteins that are recognised by the antibodies will become fluorescent, while cells lacking such proteins will not. You can then measure the fluorescence of each cell individually, sending the fluorescent cells into one container for further study, and the non-fluorescent cells into another. This works well for studying cells, which have hundreds of surface proteins for the antibodies to bind to, producing a strong fluorescence signal. FACS can also be used to sort large viruses such as the Ebola virus, but for studying smaller viruses with fewer surface proteins - like HIV - FACS is not sensitive enough. Now researchers at EMBL, ESPCI Paris, and the International AIDS Vaccine Initiative have developed a new technique for rapidly sorting HIV viruses, which could lead to more rapid development of a vaccine for HIV, as they report in Cell Chemical Biology. Study author Christoph Merten explains.
What did you do?
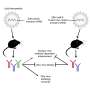
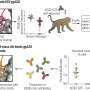
We developed a system that enables us to analyse and sort HIV at a rate of hundreds of viruses per second, separating the viruses according to whether or not their surface proteins have features recognised by specific antibodies. Instead of using fluorescent antibodies that would bind directly to the viral proteins - producing only a weak signal - we took the ordinary, non-fluorescent antibodies and attached them to an enzyme called alkaline phosphatase (AP). We then enclosed the viruses individually in droplets of liquid, along with a chemical that becomes fluorescent when acted on by AP. The antibodies bind to the viral proteins, and the attached AP enzymes produce many fluorescent molecules which remain inside the liquid droplet, creating a strong fluorescence signal. If the virus's proteins don't have the right features, the antibodies with their AP enzymes will not bind and no fluorescence is produced. We can therefore study individual viruses, sorting them with high accuracy according to whether they show features that could be exploited in developing a vaccine against HIV.
Ours is a microfluidic system - in other words, it uses technology designed for manipulating extremely small quantities of liquid. The whole system is contained on a microfluidic 'chip' - a palm-sized device consisting of microscopic networks of channels for liquid to flow through. These channels are just a few hundredths of a millimetre across, and each droplet in our experiments is around 30 billionths of a millilitre. Microfluidic chips offer particular advantages when working with pathogens like HIV, since they're completely sealed and therefore very safe to use. Typical FACS systems can produce airborne droplets, so much more stringent containment measures are required when working with harmful bacteria and viruses.
Why does it matter?
Our method makes it possible to analyse and sort HIV viruses in quantities and at speeds that have not been possible before. This enables us to rapidly test millions of viral variants, which should significantly speed up the process of vaccine development.
In our experiments each droplet contained a virus and antibodies, but it should also be possible to add a cell to the droplet and study whether the antibodies can stop the virus from entering the cell. That's not possible with FACS, so it opens up many possibilities for future research.
More information: Cell Chemical Biology (2017). DOI: 10.1016/j.chembiol.2017.05.009