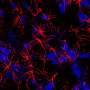
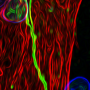
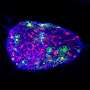
Model for producing brain's 'helper cells' could lead to treatments for Alzheimer's
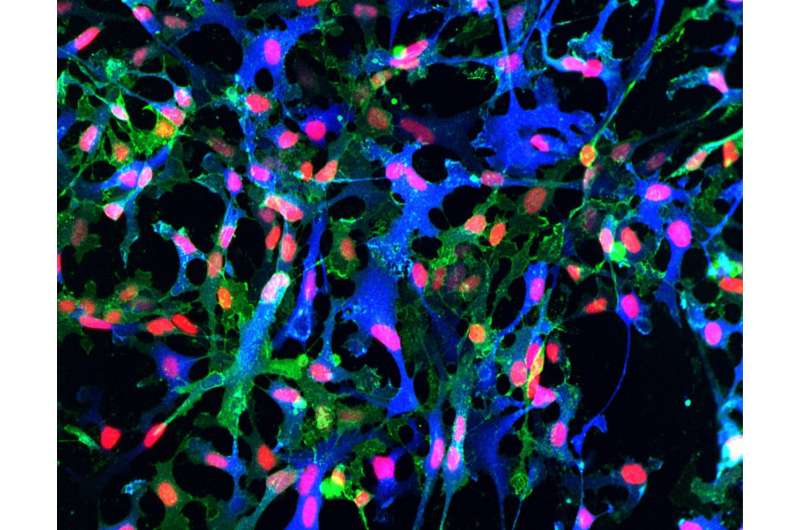
A Swedish research team has published a new protocol with the potential for industrial-scale production of the brain helper cells known as astrocytes. Their work could help medical science develop treatments for such diseases as Alzheimer's.
Star shaped cells that are found in the brain and spine, astrocytes were long thought to be the "glue" that binds nerve cell. But recent advances show that they are much more. Astrocytes are responsible for complex regulation of a variety of critical brain functions and have proven to be central to neurological diseases such as Alzheimer's.
But for research, these cells prove problematic. Astrocytes are significantly more complex than those found in mice, for example. And existing models for their production have not quite sufficed for development of drug treatments.
Drawing on Nobel Prize-winning technology, researchers from KTH Royal Institute of Technology, Karolinska Institutet and Astra Zeneca, reprogrammed human skin cells to create induced pluripotent stem cells, or iPS cells. "These cells were then guided with growth factors to become astrocytes," says the study's senior author Anna Falk, associate professor in the Department of Neuroscience at Karolinska.
Beginning with stem cells, researchers can produce an infinite number of astrocytes, which is important for the large-scale use within the pharmaceutical industry, says Anna Herland, assistant senior lecturer in the Department of Micro and Nanosystems at KTH.
"The historically high statistics of clinical failures in developing drugs against neurological diseases have now made drug companies increasingly interested in improved cell models in which human cells are used," Herland says. "Our work has been focused on development of a cell model that follows human embryonic development of astrocytes."
Lead author Anders Lundin, a PhD student at AstraZeneca in Gothenburg, says that the study compared its cellular models with those used in the pharmaceutical industry today. "We can show significant differences," he says.
The researchers' model shows wider functionality than the other models, Falk says. "According all functional and descriptive analyses we performed, the astrocytes we developed work similarly to those found in the brain."
"The other models which we compared demonstrate functions in some of the analyses. We have also done a pilot screening with a few substances but under the same conditions as a major drug screening, and it shows that our astrocyte model has the potential to identify new candidates who can go into drug development for neurological diseases," Herland says.
The researchers say the results from analysis of an astrocyte must be validated in another model in order to ensure the results are universal.
"Our model of human astrocytes is an important step forward in order to understand and attack human neurological diseases where astrocytes have an important role," she says. "With this model, we can begin to study how astrocytes develop and receive their functional diversity during embryonic development."
More information: Anders Lundin et al. Human iPS-Derived Astroglia from a Stable Neural Precursor State Show Improved Functionality Compared with Conventional Astrocytic Models, Stem Cell Reports (2018). DOI: 10.1016/j.stemcr.2018.01.021