Traffic jam causes immune cell road rage
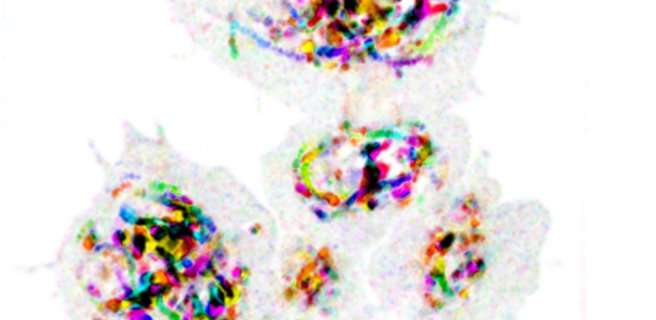
Mutations in the gene OCRL cause Lowe syndrome, but it has remained unclear how loss of OCRL leads to the diverse eye, kidney, and neurological symptoms suffered by patients. In the featured article from the October issue of PLOS Genetics, we report that loss of OCRL in fruit flies causes inappropriate activation of immune cells, raising the exciting possibility that inflammation might be a cause of some Lowe syndrome symptoms.
Flies provide fresh insight into Lowe syndrome
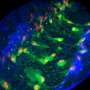
The OCRL gene encodes an enzyme that controls the composition of many types of lipid membranes in cells. A major challenge in understanding Lowe syndrome has been that OCRL appears to play several different roles in cells, including controlling how they divide, how they sense their surroundings, and how they store and transport materials in intracellular compartments called endosomes. Most experiments studying OCRL have been performed on cells grown in culture, and we reasoned that testing where and how OCRL works in an intact organism might help tease out its most relevant functions. To do this, we deleted the fruit fly homolog of OCRL, and unexpectedly found that the mutants exhibited activation of macrophage-like immune cells. These cells had many of the same problems observed in cultured cells lacking OCRL, including defects in cell division, endocytosis, and endosomal trafficking. Interestingly, we found that neither defects in cell division nor endocytosis could account for the immune cell phenotype, suggesting a problem with endosomal trafficking.
A traffic jam causes immune cell activation
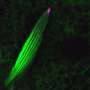
Endosomal trafficking controls the uptake, sorting, recycling, and degradation of a wide range of signals that regulate a variety of cell behaviors. This process is highly dynamic, as illustrated by the cover image from the October issue of PLOS Genetics (see also movie here). These trafficking dynamics depend on a family of proteins called Rabs, which determine the functions of different types of endosomes and control the movements of cargo between them.
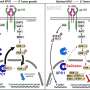
To test whether endosomal trafficking is involved in the OCRL mutant phenotype, we genetically manipulated the activity of Rab proteins specifically in immune cells. We found a primary role for Rab7 and Rab11, which are two Rabs that help sort cargo between recycling and degradative endosomes. In otherwise normal immune cells, activation of Rab7 or inactivation of Rab11 caused a phenotype similar to loss of OCRL. Further, opposite manipulations of each Rab were able to suppress immune cell activation in OCRL mutants, indicating that this sorting step is the critical site of action of OCRL in immune cells. Going forward, it will be crucial to determine how this change in traffic affects specific immune-relevant signals.
A potential link to seizures in Lowe syndrome patients?
Several lines of evidence link the immune cell activation we observed to seizures in Lowe syndrome patients. We found that OCRL mutants amplify multiple immune signals that regulate inflammatory responses in the human brain, including Toll-like receptors and IL6. Importantly, these inflammatory pathways have been implicated in seizure disorders in humans. Other groups have identified direct links between OCRL and inflammation: Loss of OCRL in zebrafish causes cystic lesions in the brain and seizure susceptibility, while induction of seizures in a mouse model of epilepsy altered OCRL levels in brain astrocytes [3, 4]. Together, these data suggest that inflammation may be a promising new avenue of investigation into the seizure symptoms of Lowe syndrome patients.
More information: Steven J. Del Signore et al. dOCRL maintains immune cell quiescence by regulating endosomal traffic, PLOS Genetics (2017). DOI: 10.1371/journal.pgen.1007052
Legido A, Katsetos CD. Experimental studies in epilepsy: immunologic and inflammatory mechanisms. Semin Pediatr Neurol. 2014;21(3):197–206. Epub 2014/12/17. pmid:25510941
Ramirez IB, Pietka G, Jones DR, Divecha N, Alia A, Baraban SC, et al. Impaired neural development in a zebrafish model for Lowe syndrome. Hum Mol Genet. 2012;21(8):1744–59. Epub 2012/01/03. pmid:22210625
Clasadonte J, Morel L, Barrios-Camacho CM, Chiang MS, Zhang J, Iyer L, et al. Molecular analysis of acute and chronic reactive astrocytes in the pilocarpine model of temporal lobe epilepsy. Neurobiol Dis. 2016;91:315–25. Epub 2016/04/10. pmid:27060558
This story is republished courtesy of PLOS Blogs: blogs.plos.org.