Adult-like human heart muscle grown from patient-specific stem cells
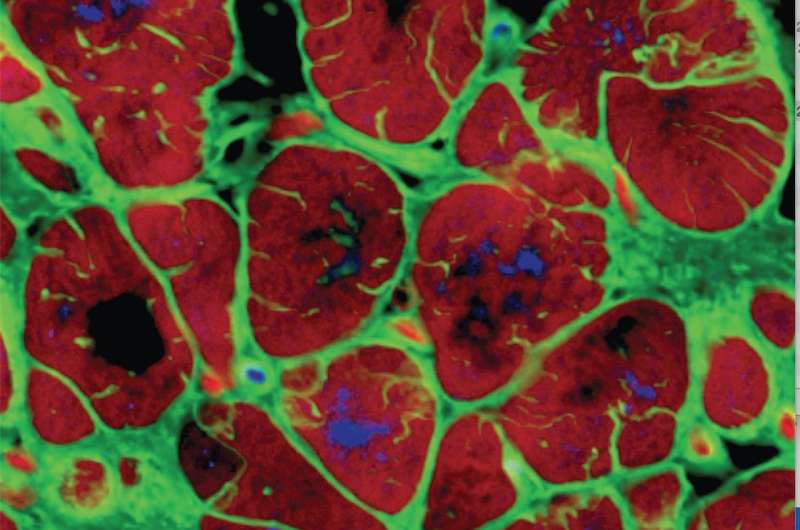
Growing human cardiac tissue that behaves like native heart muscle would be transformative for biomedical research, enabling researchers to study human physiology and model heart diseases under fully controlled conditions. While today scientists can grow many tissues, including the heart muscle, from stem cells taken from a small blood sample of any of us, current bioengineered tissues fail to show some of the most critical hallmarks of adult human heart function. Researchers have not been able to build a cultured heart muscle mature enough to be useful for medical research—until now.
Investigators at Columbia Engineering have developed a radically new approach to growing in the lab adult-like human heart muscle from blood-derived human induced pluripotent stem cells (iPSCs), over only four weeks of bioreactor cultivation. They essentially compressed the timeframe for development, which normally takes nine months, into a faster and more complete transition to cardiac maturity than any other team has been able to achieve. Their methodology is to form human cardiac tissues from early-stage iPSC-derived cardiomyocytes, soon after the initiation of spontaneous contractions, by subjecting the cells encapsulated in hydrogel to increasingly intense physical conditioning. Their findings are published today in Nature.
"Many of the ongoing efforts—including those from our lab—have been biomimetic in nature, trying to recapitulate the known events present during native development," says the study's senior author Gordana Vunjak-Novakovic, University Professor, The Mikati Foundation Professor at Columbia Engineering, and professor of medicine at Columbia University Vagelos College of Physicians and Surgeons. "Because these efforts have been limited in how much maturation can be achieved, we decided to try something totally new: to explore the concept of accelerated development. It took a lot of creative thinking and clever engineering by the whole team across both campuses of Columbia University to develop the model we now have, a highly matured, patient-specific heart muscle that can be used for studies of heart development, physiology, disease, and responses to drugs."
The team used early-stage cardiomyocytes, derived from the human stem cells obtained from blood samples that are spontaneously beating but still have a lot of developmental plasticity. They encapsulated these cardiomyocytes and the supporting cells in a fibrin gelling solution needed to form an initial tissue construct around two elastic pillars. They cultured the tissues in a multi-chamber platform—an organ on a chip device that contains numerous biological replicates of their small-size human heart muscle (measuring about 6 mm in length). In this platform, the researchers applied electrical pacing to force the bioengineered heart muscle to twitch and work against the load (by pulling the elastic pillars), which is exactly what happens in healthy heart muscle in the body.
In addition to the environmental control and the application of molecular and physical regulatory factors, the platform also enabled optical measurements of numerous functional properties. The team developed the methods and software to measure the frequency, amplitude, force of contractions, and calcium signal propagation, among other important tissue properties and responses to drugs.
"The common approach in our field has been that the more mature the starting cardiomyocytes, the better," says the study's lead author Kacey Ronaldson-Bouchard, then a graduate student and now a postdoctoral scientist in Vunjak-Novakovic's Laboratory for Stem Cells and Tissue Engineering. "However, we found that very early-stage cells, which still have developmental plasticity, would respond better to the external signals we deliver to drive maturation."
The other major advance in this study is that, instead of the gentle mechanical stretch that is present in a developing fetal heart, the researchers applied a special regime of electromechanical conditioning, by increasing the frequency of electrically induced contractions little by little every day. This regime forced the cultured muscle to work?each day harder than the last. The goal was to see if the bioengineered heart would respond like a native heart by gradually accommodating the increased load and making the fetal to postnatal transition.
The technique worked. The researchers saw comprehensive changes at all levels, leading to rapid and unprecedented maturation of the tissue structure, metabolism, and function. Over just four weeks of culture, the tissues displayed adult-like gene expression profiles, remarkably organized ultrastructure, and a number of functional features seen in the mature human heart muscle. These included the physiologic length of the sarcomere (contractile machinery of the cell); the physiologic density of mitochondria (energy factory of the cell); the presence of transverse tubules (critical, previously undocumented features of the cell membrane that help the cell respond quickly to changes in calcium and transmit signals); and the switch to oxidative metabolism and functional calcium handling.

Vunjak-Novakovic's team has shown, both in this study and other recent work, that they can use the matured human heart muscle to recapitulate the phenotype of some heart conditions, such as the pathological hypertrophy of the heart or the reduced contractility associated with calcium channel mutations. They are extending these studies into broader aspects of disease modeling to get a better understanding of the mechanistic basis of cardiac disease and cardiotoxicity induced by drugs used to treat other organ systems. Their work could facilitate discovery of new therapeutic targets and lead to new cardioprotective or curative treatment modalities. This ongoing research is part of the "organs on a chip" project funded by the National Institutes of Health, which uses multi-tissue platforms that include the adult heart muscle referenced in this paper as well as bone, liver, vasculature, skin, and solid tumors.
"The resulting engineered tissue is truly unprecedented in its similarity to functioning human tissue," said Seila Selimovic, director of the NIBIB (National Institute of Biomedical Imaging and Bioengineering) Tissue Chips program, within the National Institutes of Health that funded this research. "The ability to develop mature cardiac tissue in such a short time is an important step in moving us closer to having reliable human tissue models for drug testing." The better the engineered tissues emulate the human heart, the better they can predict the effects that drugs or environmental factors have on the actual heart tissue of a patient. Having a reliable human tissue model would help make drug development significantly faster, safer and cheaper.
More information: Kacey Ronaldson-Bouchard et al, Advanced maturation of human cardiac tissue grown from pluripotent stem cells, Nature (2018). DOI: 10.1038/s41586-018-0016-3