Defects in tissue trigger disease-like transformation of cells
Homeowners know that one little termite can lead to big problems: while termites are efficient at gnawing away at wood, they can do even more damage if the wood is already broken or has another defect.
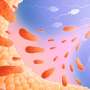
Mechanical engineers at Washington University in St. Louis have found the same effect in some of the body's tissue: One small defect in tissue boundaries known as the basement membrane can lead normal cells to take on characteristics of diseased cells, such as cancer cells, and invade the surrounding tissue.
In research published online May 16 in Integrative Biology, Amit Pathak, assistant professor of mechanical engineering in the School of Engineering & Applied Science, and his team found this feedback loop with epithelial cells. These simple cell colonies act as the body's defense against the outside world and also line the inside of the throat, intestines, blood vessels and organs, meaning the epithelial cells could provide a little link to a much bigger issue. Knowledge of this relationship could help researchers find new therapies to halt cancer metastasis.
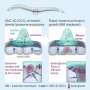
Researchers already knew that in cancer, epithelial cells transition into mesenchymal types that can degrade the basement membrane, a process stimulated by enzymes called matrix metalloproteinases (MMPs). This process causes further defects in healthy tissue and allows cancer to metastasize. Pathak's team is the first to discover that the reverse also is true—pre-existing defects in the basement membrane, such as a wound or small incision, are enough to encourage the normal cells to take on disease characteristics, leading to further degradation of the basement membrane.
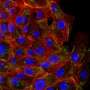
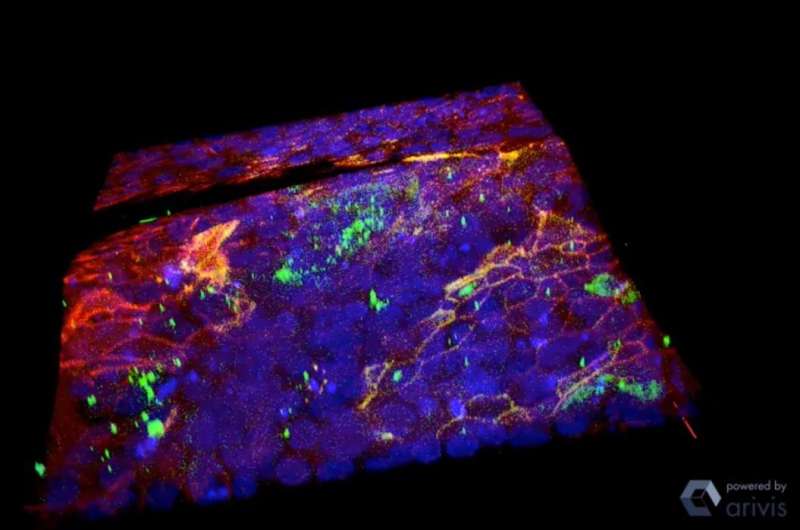
Pathak and his team fabricated a basement membrane model using a hydrogel coated with collagen IV, a protein that is the main and most important structural component of the basement membrane. They added normal epithelial cells, which like to stick together, then made a small cut in the gel. After six days, the cells started to break apart, move around and invade the gel, mimicking a tumor invading healthy tissue.
"Because of the defect, the cells started to show signatures of diseased cells," Pathak said. "If there was no defect, they would have stayed the same. This small defect of less than one-tenth of a millimeter caused this whole process of these cells changing."
To show that the cells would have remained the same without the defect, Pathak and his team treated them with an inhibitor of MMPs, which are known to degrade the basement membrane in wound healing. The cells did not change and did not invade the basement membrane.
Previously, researchers believed that once tumor cells became malignant, they would degrade the basement membrane.
"This finding shows that we shouldn't take basement membrane degradation as an inevitable outcome," Pathak said. "In addition to targeting tumor cells, we can determine therapeutic strategies that make the basement membrane more stable. If it is more stable, then we can at least slow down and possibly reverse metastasis."
In addition to the hydrogel model, Pathak and his team made a computational model integrating the biochemical and biophysical effects of the defects on the cellular transitions, which allowed them to make predictions on which they will base future experiments.
More information: Christopher Walter et al. Physical defects in basement membrane-mimicking collagen-IV matrices trigger cellular EMT and invasion, Integrative Biology (2018). DOI: 10.1039/C8IB00034D