New therapeutic opportunity for the treatment of resistant malignant melanoma
A team of researchers led by Dr. Pierre Close, WELBIO researcher at the ULiège GIGA Institute and Dr. Francesca Rapino has uncovered a new therapeutic opportunity in the treatment of malignant melanoma that has acquired resistance to targeted therapies. In collaboration with researchers from VIB, they have revealed that malignant melanoma cells can reprogram their protein synthesis machinery and become addicted to a new family of enzymes that modify transfer RNAs during acquired resistance. Strikingly, the inhibition of these molecules with targeted therapies produces a strong anti-tumoral effect. These new findings, published in Nature, will contribute to the development of improved diagnostic tools and melanoma treatment.
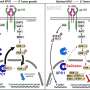
Resistance to therapy inhibits the effectiveness of current treatments for aggressive cancers such as malignant melanoma. The onset of resistance relates to the capability of tumor cells to circumvent the stress induced by the treatment. In order to survive, cancer cells develop a series of adaptive mechanisms by rewiring fundamental processes. Among those, reprogramming of mRNA translation favors the expression of proteins essential for tumor development. The lab of Dr. Close has been studying the contribution of wobble tRNA modification in cancer development through regulation of selective mRNA translation for a few years now, uncovering their central role in tumor initiation and metastatic potential.
Modification of certain tRNA molecules at the wobble position regulates selective mRNA translation and impact on protein expression. The GIGA-ULiège team discovered that melanoma that carry the BRAF(V600E) mutation—found in more than 50 percent of the melanoma patients—are addicted to enzymes modifying wobble uridine tRNAs (U34-tRNA).
Francesca Rapino, post-doctoral researcher in Dr. Close's lab, says, "Our research showed that wobble uridine tRNA modification enzymes are upregulated in melanoma clinical samples and have low expression in melanocytes, the normal melanin-producing cells. Strikingly, inhibition of this family of enzymes led to a very strong and specific cell death in BRAF(V600E) melanoma, but had no effect on melanocytes. This very specific effect led us to postulate that these enzymes may play an important role in melanoma development."
Growing melanoma tumors adapt their metabolism and use glucose (sugar) as source of energy. The scientists from the University of Liège demonstrated that U34-tRNA enzymes are key for the expression of proteins involved in glucose metabolism.
Pierre Close (WELBIO, ULiège), says, "Using melanoma-derived samples, we found that U34-tRNA enzymes are essential to sustain glucose metabolism. Therefore, the inhibition of these enzymes prevents glucose metabolism in melanoma cells, and limits their energy income. As a consequence, the growth and survival of melanoma cells is strongly reduced after inhibition of U34-tRNA enzymes. Strikingly, we discovered that acquired resistance to targeted therapy, which strongly limits the clinical benefit of the treatment of malignant melanoma, is prevented by the inhibition of U34-tRNA enzymes. In other words, the inhibition of these enzymes synergizes with targeted therapies to block malignant melanoma growth."
This work revealed the clinical potential of U34-tRNA enzymes inhibition for the treatment of human malignant melanoma, a disease that remains extremely difficult to treat. Further research will be necessary to firmly establish the real clinical benefit of this approach and to develop therapeutic tools that could achieve this goal.
More information: Francesca Rapino et al, Codon-specific translation reprogramming promotes resistance to targeted therapy, Nature (2018). DOI: 10.1038/s41586-018-0243-7