Connective tissue on the wrong road—When organs start to scar

Scientists from Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU), Department of Medicine 3—Rheumatology and Immunology, headed by Prof. Dr. Georg Schett, have now decrypted a molecular network that could in future provide a new way to treat organ scarring. The results show that the protein PU.1 causes pathological deposition of connective tissue. The scientists have now published their results in Nature.
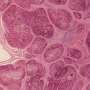
In connective tissue diseases such as systemic sclerosis, referred to collectively as fibrosis, excessive activation of connective tissue cells leads to hardening of the tissue and scarring within the affected organ. In principle, these diseases can affect any organ system and very often lead to disruption of organ function. Connective tissue cells play a key role in normal wound healing in healthy individuals. However, if the activation of connective tissue cells cannot be switched off, fibrotic diseases occur in which an enormous amount of matrix is deposited in the tissue, leading to scarring and dysfunction of the affected tissue. Until now, scientists did not fully understand why repair processes malfunction in fibrotic diseases.
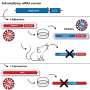
An international team of scientists led by Dr. Andreas Ramming from the Chair of Internal Medicine III at FAU has now been able to decipher a molecular mechanism responsible for the ongoing activation of connective tissue cells. In experimental studies, the researchers targeted the protein PU.1. In normal wound healing, the formation of PU.1 is inhibited by the body so that at the end of the normal healing process the connective tissue cells can return to a resting state.
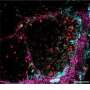
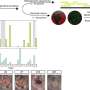
"We were able to show that PU.1 is activated in various connective tissue diseases in the skin, lungs, liver and kidneys. PU.1 binds to the DNA in the connective tissue cells and reprogrammes them, resulting in a prolonged deposition of tissue components," explains Dr. Ramming.
PU.1 is not the only factor involved in fibrosis, as factors that are involved in the deposition of scar tissue have already been identified in the past. What has been discovered now, however, is that PU.1 plays a central role in a network of factors controlling this process. "PU.1 is like the conductor in an orchestra," explains Ramming. "If you take it out, the entire concert collapses." This approach has already been tested using an experimental drug, fueling the hope that clinical trials on inhibiting PU.1 may soon be able to be launched, aimed at treating fibrosis better.
More information: Thomas Wohlfahrt et al, PU.1 controls fibroblast polarization and tissue fibrosis, Nature (2019). DOI: 10.1038/s41586-019-0896-x