In severe COVID, cytokine 'hurricane' in lung attracts damaging inflammatory cells
A cytokine 'hurricane' centered in the lungs drives respiratory symptoms in patients with severe COVID-19, a new study by immunologists at Columbia University Vagelos College of Physicians and Surgeons suggests.
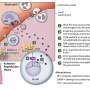
Two cytokines, CCL2 and CCL3, appear critical in luring immune cells, called monocytes, from the bloodstream into the lungs, where the cells launch an overaggressive attempt to clear the virus.
Targeting these specific cytokines with inhibitors may calm the immune reaction and prevent lung tissue damage. Currently, one drug that blocks immune responses to CCL2 is being studied in clinical trials of patients with severe COVID-19.
Survivors of severe COVID-19, the study also found, had a greater abundance of antiviral T cells in their lungs than patients who died, suggesting these T cells may be critical in helping patients control the virus and preventing a runaway immune response.
The study, published online March 12 in the journal Immunity, is one of the first to examine the immune response as it unfolds in real time inside the lungs and the bloodstream in patients who are hospitalized with severe COVID-19.
Treatments for Severe COVID-19 Needed
In patients with severe COVID-19, the lungs are damaged, and patients need supplemental oxygen. The risk of mortality is over 40%.
"We wanted to look at the immune response in the lung in severe disease, because it's those responses that are either protecting the organ or causing the damage," says Donna Farber, Ph.D., professor of microbiology & immunology and the George H. Humphreys II Professor of Surgical Sciences in the Department of Surgery, who led the study. "Even though individuals are getting vaccinated, severe COVID-19 remains a significant risk for certain individuals and we need to find ways to treat people who develop severe disease."
Numerous COVID studies have focused on identifying immune responses in blood; a few have looked at airway samples from a single time point or from autopsies. Few studies have examined the immune response to SARS-CoV-2 in the respiratory tract as the response unfolds, because obtaining such samples from patients is challenging. But the Columbia researchers learned several years ago that they can retrieve respiratory immune cells from the routine daily saline washes of the endotracheal tubes that connect intubated patients to a ventilator.
Paired Airway and Blood Samples Show Complete Immune Response in Real Time
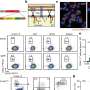
In this new study, the researchers collected respiratory immune cells from 15 COVID-19 patients who had been intubated. Each patient spent four to seven days on a ventilator, and airway and blood samples were obtained daily.
All the samples were examined for the presence of cytokines and different types of immune cells. For four of the patients, the researchers measured gene expression in every immune cell to get a detailed picture of the cells' activities.
"It seems obvious that the immune response in the respiratory tract would drive a disease caused by a respiratory virus, but we didn't know what the processes were and how they worked together with systematic responses," Farber says. "What's new here is that we've been able to simultaneously sample both the respiratory tract and the blood over time and put together a more complete picture of the responses involved and how local and systemic responses work together."
Two Cytokines Appear to Drive Lung Damage
Though the researchers found elevated levels of many cytokines in the blood, many more types of cytokines were present in the lungs and at highly elevated levels.
"People refer to patients experiencing a cytokine storm in the blood, but what we're seeing in the lungs is on another level," Farber says. "The immune cells in the lung went into overdrive releasing these cytokines."
No cytokines were found in the blood that weren't also found in the lung, suggesting that the signals causing the severe inflammation are driven by lung cytokines rather than systemic ones.
"It has been suggested that systemic cytokines are driving severe disease, but our results suggest that inflammatory processes that perpetuate disease are coming from the lungs," Farber says.
CCL2 and CCL3 cytokines released by the lung appeared to be particularly important in severe disease, because the monocytes drawn into the lung expressed receptors for these molecules. "Normally, these cells never make it to the airway, but in severe COVID patients, they accumulate throughout the lung and clog up the alveolar spaces," Farber says.
The findings also may explain why trials of other cytokine inhibitors, including tocilizumab, have shown variable efficacy. Tocilizumab inhibits the cytokine IL-6, which is elevated in patients with severe COVID but does not appear to be a major component of inflammation in the lung, Farber says.
Survivors Have High Level of T Cells in Lung
Of the study's 15 patients, eight died and all survivors were under 60 years of age.
The lungs of those survivors had significantly more T cells, which are mobilized to the lung to clear virus, and a lower proportion of inflammatory macrophages and monocytes.
In general, younger people have a more robust T cell response while older people have a higher baseline level of inflammatory cells; both factors may help explain why older patients with severe COVID fare worse.
The cell differences between patients who lived and those who died could potentially lead to a way to predict which patients are more likely to develop severe disease, although the differences are only apparent in the lung, not the blood. Importantly, the predictive value of airway immune cell frequencies was better than standard clinical measurements of lung and organ damage.
"Our next step is to try to find a more accessible biomarker that predicts severe COVID so we can try to give treatments earlier to patients who are most at risk," Farber says.
"Understanding the immune response in severe COVID is really critical at this point," Farber adds, "because we could see this again with the next coronavirus outbreak. This is what coronaviruses do at their worst; this is their M.O."
More information: Peter A. Szabo et al, Longitudinal profiling of respiratory and systemic immune responses reveals myeloid cell-driven lung inflammation in severe COVID-19, Immunity (2021). DOI: 10.1016/j.immuni.2021.03.005