Exploring autoimmunity's regulatory roots
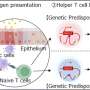
Our genome contains vast networks that regulate the activity of protein-coding genes. Variations in this regulatory DNA (which does not encode proteins) may affect both when and how much of a given protein a cell produces. Disruptions in regulation can have a profound impact on healthy function—in fact, most genomic variants discovered by genome-wide association studies (GWAS), which identify variant locations within the genome associated with susceptibility to disease, are found in non-coding regions.
GWAS provides a good starting point for finding genetic associations with disease, but can implicate dozens or even hundreds of variants for every actual causal variant, so prioritizing them is essential. A team of researchers from The Jackson Laboratory (JAX), Benaroya Research Institute, and the Broad Institute of MIT and Harvard paired two high-throughput methods to examine GWAS-defined variants associated with five autoimmune diseases. In the process, they were able to zero in on non-coding variants associated with autoimmune disorders, one of which appears to play a key role in keeping the immune system's T cells in check. Their findings appear in the journal Nature Genetics.
From disease association to functional disruption
The research team, led by associate professor Ryan Tewhey of JAX, assistant member John Ray of Benaroya Research Institute, and institute member Nir Hacohen of the Cell Circuits Program at Broad, applied two methods—massively parallel reporter assays (MPRA) and chromatin accessibility studies—to analyze GWAS data from studies of type 1 diabetes, rheumatoid arthritis, ulcerative colitis, Crohn's disease, and multiple sclerosis. MPRA tests variants for their ability to affect gene expression, while chromatin accessibility data show which areas of the genome are likely to be accessible to protein binding and involved with gene expression.
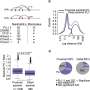
"Both MPRA and chromatin accessibility data are helpful for narrowing down the GWAS variants actually associated with disease, but alone they don't provide sufficient precision," said Tewhey, who worked on MPRA approaches while a postdoctoral fellow at Broad. "By combining them, we were able to enrich our selection of the actual causal variants nearly 60-fold and identify single variants associated with multiple autoimmune diseases."
Starting with a list of approximately 18,000 variants, the team was able to home in on 60 that likely have a causal relationship with autoimmunity. Because one variant, known as rs72928038, was associated with multiple autoimmune disorders, the researchers decided examine it further by engineering it into human T cells in vitro. They found that in the engineered cells the variant reduced the expression of the gene BACH2, which regulates T cells' differentiation into different subclasses with different functions.
A notable change in mice
Because genomic region containing rs72928038 is highly conserved between humans and mice, Tewhey, Ray, Hacohen, and their colleagues—including study co-first authors Kousuke Mouri of JAX and Michael Guo of Broad—then used a mouse model system to understand how the variant may influence disease progression. They found that deleting a small portion of the genome containing rs72928038 again reduced BACH2 expression in mice. In addition, the variant appeared to play an important role in suppressing naive T cell activation, keeping the cells from becoming activated before being directed toward a specific pathogen or non-tissue target. That finding likely explains the variant's association with autoimmune diseases.
"Not many mouse models have been engineered to study non-coding variants associated with disease," said Tewhey. "It's risky, because the effect size can be so small in isolation that it doesn't change the physiology of the mouse.
"The deletion we made for the study only reduced Bach2 gene expression by about 20 percent, but fortunately it produced a phenotype relevant to autoimmunity," Tewhey continued. "The results provide a good proof point for expanding the use of mice to investigate non-coding variants associated with human disease identified by GWAS."
More information: Kousuke Mouri et al, Prioritization of autoimmune disease-associated genetic variants that perturb regulatory element activity in T cells, Nature Genetics (2022). DOI: 10.1038/s41588-022-01056-5