This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Q&A: Illuminating the mechanisms behind severe inflammation in asthma
Joshua A. Boyce, MD, chief of the Division of Allergy and Clinical Immunology at Brigham and Women's Hospital, is the senior author of a paper published in Immunity, "Mast Cells Control Lung Type 2 Inflammation via Prostaglandin E2-Driven Soluble ST2."
How would you summarize your study for a lay audience?
Previous research indicates that mast cells, a type of immune cell found in the tissues throughout the body, cause inflammation during an asthma attack, inducing symptoms such as trouble breathing and narrowing of the airways.
Our study demonstrates that prostaglandin E2 (PGE2), a naturally occurring chemical in the human body previously thought to inhibit mast cell activation, elicits mast cells to prevent inflammation by creating a molecule called soluble ST2 (sST2), which can mitigate signs and symptoms of asthma by blocking the actions of interleukin 33, a powerful proinflammatory cytokine.
What knowledge gaps does your study help to fill?
Our work suggests that mast cells, rather than simply being drivers of inflammation, can also mitigate inflammation when PGE2 is present. It also suggests that PGE2 actively enlists mast cells to suppress the inflammation that leads to asthma exacerbations.
How did you conduct your study?
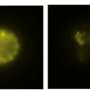
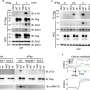
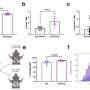
We utilized a variety of methods during our study. Using mouse models of airway inflammation, we examined how deficiencies in PGE2, the loss of PGE2 receptors from mast cells, and the absence of ST2 from mast cells impacted the severity of inflammation.
We also found that the levels of PGE2 metabolites in the urine correlated strongly with serum sST2 levels in patients with severe asthma and nasal polyps, and that high serum sST2 levels were associated with milder disease and better lung function. We also determined that PGE2, rather than simply inhibiting mast cell activation, profoundly alters gene expression by mast cells using a technique known as RNA sequencing.
What are the implications?
Through our work, we found that mast cells likely protect against the mechanisms that drive severe inflammation under conditions of normal PGE2 generation. However, when PGE2 isn't produced sufficiently, which is common in severe asthma and nasal polyposis, the lack of mast cell-derived sST2 contributes to the severity of disease.
Our findings suggest a promise in targeting PGE2 with therapeutics to alter the levels of sST2 in patients with severe asthma. They also suggest that antibodies against ST2 and IL-33, currently in clinical trials. may work best in patients with the lowest levels of PGE2 production.
What are the next steps?
We plan to test the hypothesis that PGE2 and sST2 levels can predict the efficacy of new medicines, including monoclonal antibodies, developed for the treatment of severe asthma and nasal polyposis.
In addition to Boyce, additional Brigham and Women's Hospital authors include Kinan Alhallak, Jun Nagai, Kendall Zaleski, Sofia Marshall, Tamara Salloum, Tahereh Derakhshan, Hiroaki Hayashi, Chunli Feng, Radomir Kratchmarov, Juying Lai, Virinchi Kuchibhotla, Airi Nishida, Barbara Balestrieri, Tanya Laidlaw, Daniel F. Dwyer.
More information: Mast Cells Control Lung Type 2 Inflammation via Prostaglandin E2-Driven Soluble ST2., Immunity (2024). DOI: 10.1016/j.immuni.2024.05.003. www.cell.com/immunity/fulltext … 1074-7613(24)00254-1