Cellular bells: Key step in manufacture of red blood cells decoded
A healthy adult must generate as many as one hundred billion new red blood cells each day, to maintain the numbers circulating in his blood. A team of EPFL researchers has identified a key step in the process by which red blood cells are born. The discovery could not only shed light on the causes of blood disorders such as anaemia, it could also bring closer the medics' dream of being able to manufacture red blood cells in the lab—thus providing a potentially inexhaustible supply of an essential component of blood for transfusion.
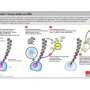
A red blood cell, or erythrocyte, is essentially a sack of haemoglobin that transports oxygen around the body. It starts life in the bone marrow as a haematopoietic stem cell, and undergoes a highly controlled process of proliferation and differentiation before acquiring its final identity.
One key step in that differentiation process is mitophagy, the elimination by absorption of the stem cell's respiratory apparatus, mitochondria. With the mitochondria gone, the cell's carrying capacity for haemoglobin is maximised. But the mechanism controlling mitophagy has never been properly understood, until now.
In a paper published this week in Science, Isabelle Barde of the EPFL's School of Life Sciences and Frontiers in Genetic Programme, and colleagues, describe experiments which show that KRAB-containing zinc finger proteins, working in concert with a cofactor called KAP1, modulate mitophagy in subtle and sophisticated ways.
The senior author on the paper, virologist Didier Trono, has been interested in the KRAB/KAP1 system for several years. 350 million years old, it is known to have a role in "silencing" components of the mammalian genome known as retroelements, because they were originally retroviruses that became incorporated into the genetic code of the organisms they infected. "It did such a good job that over the course of evolution it got co-opted to do many other things," Trono says.
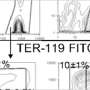
Among the roles the KRAB/KAP1 system took on was regulating mitophagy. The researchers found that mice genetically modified to lack KAP1 quickly became anaemic because they were unable to make red blood cells. More specifically, they found, the process of stem cell differentiation stalled at the stage where mitochondria were degraded in erythroblasts, the precursors of erythrocytes. And knocking out KAP1 had a similar effect in human blood cells, indicating that its role in regulating mitophagy has been conserved throughout evolution, from mouse to man.
The researchers went on to show that the KRAB/KAP1 system works by repressing repressors of mitophagy. In other words, like any good double negative, it activates the target process. That suggests that mutations in the various components of this regulatory system could contribute to blood disorders such as anaemia and certain types of leukaemia, which in turn indicates future therapeutic targets for those diseases. It also suggests ways in which red blood cell synthesis might be emulated in the lab.
But the finding has broader significance too. Mitochondria, while essential for the healthy functioning of many cells, can also be lethal to cells if they generate damaging free radicals—by-products of cellular respiration under certain conditions. The oxidative stress these free radicals produce has been implicated in liver disease, heart attacks and obesity. Hence, understanding how mitophagy is controlled could lead to a better understanding, and potentially better treatment, of those conditions.
Trono thinks that the principle of multilayered and combinatorial regulation may apply to a wide range of physiological systems. "It gives a tremendous level of modularity to nature to accomplish physiological events," he says, likening it to the way in which a pipe organ works.
An organist has both a keyboard and a pedalboard at his disposal, and he uses them in multifarious combinations to modulate the sound his instrument produces. Similarly, tiny adjustments in one or a few controlling elements can produce significant effects in many biological processes. And though mutations in any one of them could potentially lead to malfunction, the damage tends to be limited because the contribution of each one is small. That, in turn, renders the system robust. It's that robustness, Trono believes, that evolution has been selecting and refining for hundreds of millions of years.