Killing the unkillable cancer cells

Countless people are affected by the battle against cancer. Modern treatments can be quite efficient at shrinking the tumor, but too often, they can't kill all the cells, and the cancer may return. With some aggressive types of cancer, the problem is so great that there is very little that can be done for the patients.
Why do some cancer cells evade therapy? To understand this problem, researchers need to look at the molecular machinery inside the cancer cells that regulates their ability to grow, stay alive and divide, producing more cells. Scientists have identified many mechanisms within this machinery and designed drugs and other treatments that interfere with them, blocking their ability to divide, or even killing them. Clearly, these treatments are not able to kill all the cells within a tumor. This must mean that some cancer cells can use alternative mechanisms not addressed by current treatments. In this way, they evade therapy and regenerate the tumor, leading to recurrence.
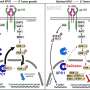
The lab of Andreas Androutsellis-Theotokis at the Department of Internal Medicine III (MK3) at the Technische Universität Dresden, in Germany previously discovered a new molecular mechanism that regulates regeneration of various tissues after injury, the STAT3-Ser/Hes3 signaling axis, named after two of its key components, and they demonstrated that it is very important in activating stem cells that live within tissues. Stem cells may help tissue regeneration in models of Parkinson's disease, ischemic stroke, multiple sclerosis, and types 1 and 2 diabetes. The researchers posited that the same mechanism could be hijacked by some cancer cells (perhaps the elusive cancer stem cells) as a means of evading current therapies and regenerating the tumor.
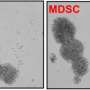
Research Fellow Steve Poser and colleagues set out to test the idea using cells from multiple patients with aggressive brain cancer. They demonstrated that these cells do switch over from using the more established molecular mechanisms to using this new mechanism. Depending on which mechanism they were using, they were vulnerable to different treatments.
The team established an international and interdisciplinary collaborative project to characterize this new cancer cell state in terms of what genes are turned on and off, what mechanical properties are affected (these are important in metastasis because they regulate how cells move and spread within the tissue), and to find treatments that kill the cells specifically when this new molecular mechanism is in use. In this way, they blocked the cancer cells from this escape route. The findings are published in the Journal of the Federation of American Societies for Experimental Biology.
The researchers identified several treatments that kill the cancer cells when they exploit this mechanism. In fact, several treatments are so specific that they kill the cells only when they operate the new mechanism and have minimal effects when they operate the more established, classical cancer growth mechanisms. Many of the treatments are currently approved for use in different indications (not cancer) and so they could potentially be repurposed in oncology.
Other treatments, such as the direct targeting of the gene Hes3 by RNA interference methods, would be new to medicine. To expedite the development of such potential therapies, the Technische Universität Dresden and collaborating institutions have formed a spinoff venture, Innate Repair. Whereas the initial focus is on aggressive brain cancer, there is already ample evidence that the same mechanism operates in many additional types of cancer. The work provides a new logic and method to identify treatments that kill these hard-to-kill cancer cells.
More information: Steven W. Poser et al. Controlling distinct signaling states in cultured cancer cells provides a new platform for drug discovery, The FASEB Journal (2019). DOI: 10.1096/fj.201802603RR